Chapter 18: Q27P (page 458)
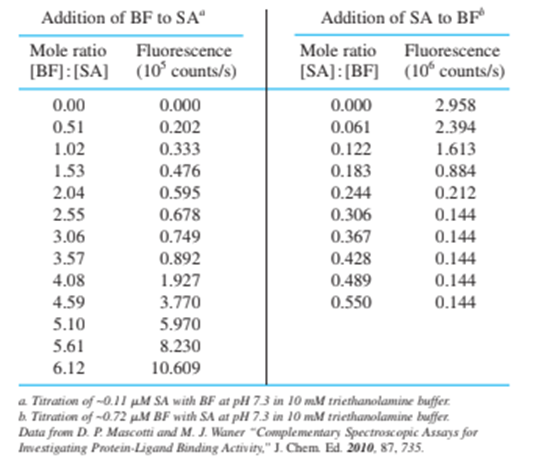
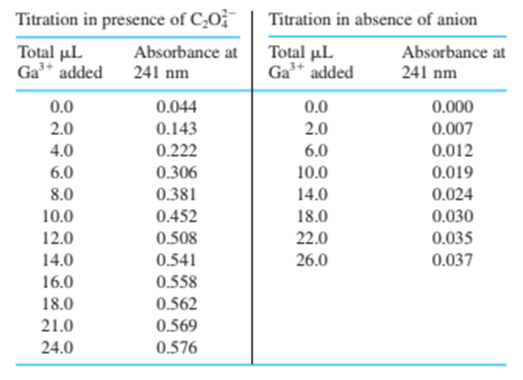
18-27. The iron-binding site of transferrin in Figure 18-8 can accommodate certain other metal ions besides Fe and certain other anions besidesData are given in the table for the titration of transferrin ( 3.57 mg in 2.00 mL ) with 6.64 mM solution in the presence of the anion oxalate, , and in the absence of a suitable anion. Prepare a graph similar to Figure 18-11, showing both sets of data. Indicate the theoretical equivalence point for the binding of one and two ions per molecule of protein and the observed end point. How many ions are bound to transferrin in the presence and in the absence of oxalate?
Short Answer
The ions that are bound to transferrin in the absence of oxalate is zero because the slope does not change



 Pyrazine
Pyrazine