Chapter 12: Q12C E (page 282)
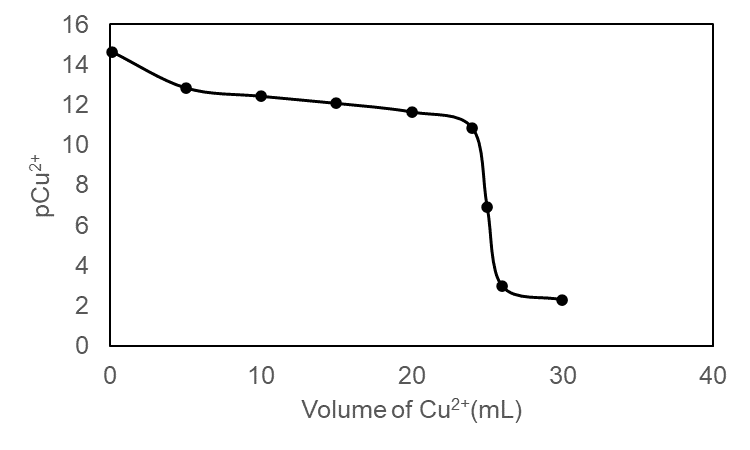
Calculate pCu2+ (to the 0.01 decimal place) at each of the following points in the titration of 50.0 mL of 0.040 0 M EDTA with 0.080 0 M Cu (NO3)2 at pH 5.00: 0.1, 5.0, 10.0, 15.0, 20.0, 24.0, 25.0, 26.0, and 30.0 mL. Make a graph of pCu2+ versus volume of titrant.
Short Answer
The values obtained forpCu2+are tabulated below
Volume (mL) | pCu2+ |
0.1 | 14.64 |
5.0 | 12.84 |
10.0 | 12.42 |
15.0 | 12.07 |
20.0 | 11.64 |
24.0 | 10.86 |
25.0 | 6.91 |
26.0 | 2.98 |
30.0 | 2.30 |
Graph of pCu2+ versus volume of titrant is plotted below