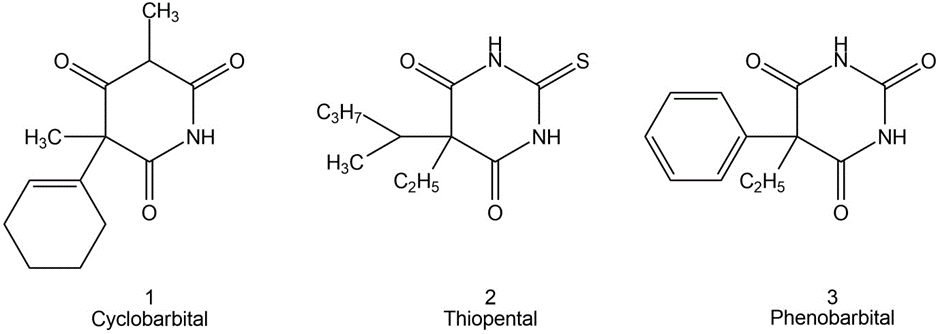
Chapter 26: Q10P (page 747)
In ion-exclusion chromatography, ions are separated from nonelectrolytes by an ion-exchange column. Nonelectrolytes penetrate the stationary phase, whereas ions of the same charge as the resin are repelled by the fixed charges. Because co-ions have access to less of the column volume, electrolytes are eluted before nonelectrolytes. The chromatogram shows the separation of trichloroacetic acid (TCA,), dichloroacetic acid (DCA,), and monochloroacetic acid (MCA,) by passage through a cation-exchange resin eluted with 0.01 M HCl. Explain why the three acids are separated and why they emerge in the order shown.

Short Answer
Ion-exclusion chromatography: Ion exclusion chromatography is used for the separation of low molecular weight ions and some natural substances by a combination of partition, adsorption, and ion repulsion.