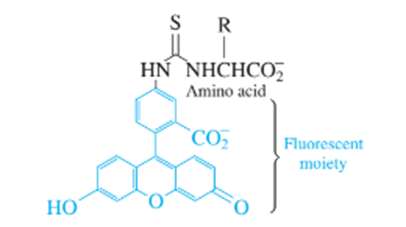
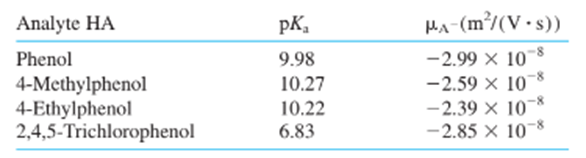
Chapter 26: Q15 P (page 747)
(a) The suppressor in Figure 26-9 enables low parts-per-billion conductivity detection for anions such as.but very poor detection limits for anions such asand borate. Explain why.
(b) Mixtures of sodium carbonate and bicarbonate can be used as eluent in suppressed-ion anion chromatography. Detection limits are poorer than with hydroxide eluent due to higher background conductivity. Explain why
Short Answer
The explanations to the above questions are:
(a). The detection limits for anions such asand borate are poor.
(b).The detection limits are poorer than with hydroxide eluent.