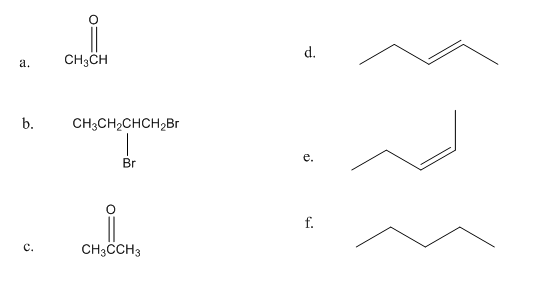
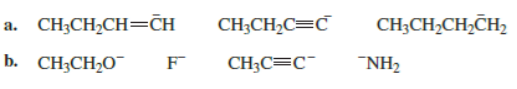Chapter 7: Q39P (page 315)
What is the major product of the reaction of 1 mol of propyne with each of the following reagents?
a.HBr (1 mol)
b.HBr (2 mol)
c.Br2(1 mol)/CH2Cl2
d.Br2 (2 mol)/CH2Cl2
e.aqueous H2SO4, HgSO4
f.R2BH in THF followed byH2O2/HO- /H2O
g.excess H2, Pd/C
h.H2/Lindlar catalyst
i.sodium amide
j.the product of part i followed by1-chloropropane