Chapter 6: Q37P (page 265)
What stereoisomers are obtained from each of the following reactions?

Short Answer
Here we have to draw the stereoisomers of the following reactions:
a)
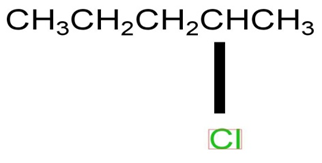
b)
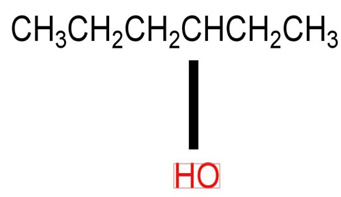
c)
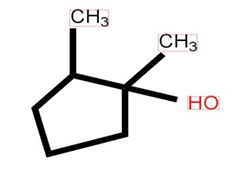
d)
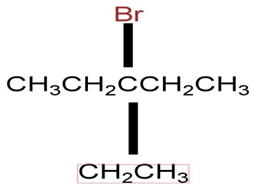
Learning Materials
Features
Discover
Chapter 6: Q37P (page 265)
What stereoisomers are obtained from each of the following reactions?

Here we have to draw the stereoisomers of the following reactions:
a)

b)

c)

d)

All the tools & learning materials you need for study success - in one app.
Get started for free
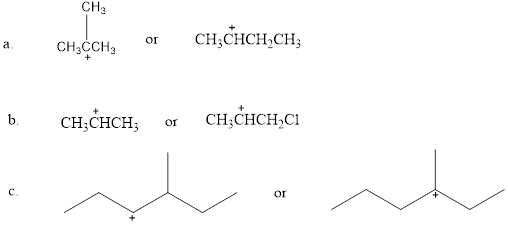
Which is more stable?
What stereoisomers are obtained from hydroboration–oxidation of the following compounds?Assign an Ror Sconfiguration to each asymmetric center.
a. cyclohexene c. cis-2-butene
b. 1-ethylcyclohexene d. (Z)-3,4-dimethyl-3-hexene
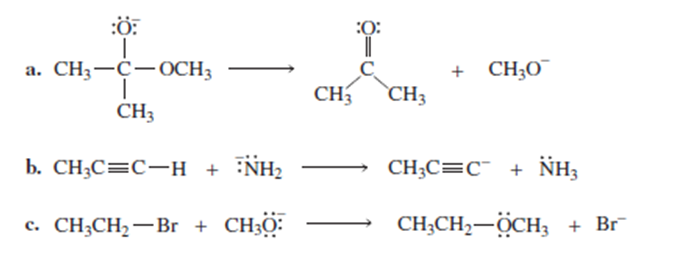
Draw curved arrows to show the flow of electrons responsible for the conversion of the following reactants into products.
Which electrophilic addition reactions
a. form a carbocation intermediate? c. form a three-membered ring intermediate?
b. form no intermediate? d. form a five-membered ring intermediate?
a. What is the major product obtained from the reaction of propene and Br2plus excess Cl-?
b. Indicate the relative amounts of the stereoisomers that are obtained.
What do you think about this solution?
We value your feedback to improve our textbook solutions.