Chapter 20: Q4P (page 954)
a. Are D-erythrose and L-erythrose enantiomers or diastereomers? b. Are L-erythrose and L-threose enantiomers or diastereomers?
Short Answer
a. Enantiomers
b. Diastereoisomers
Learning Materials
Features
Discover
Chapter 20: Q4P (page 954)
a. Are D-erythrose and L-erythrose enantiomers or diastereomers? b. Are L-erythrose and L-threose enantiomers or diastereomers?
a. Enantiomers
b. Diastereoisomers
All the tools & learning materials you need for study success - in one app.
Get started for free
a. What other monosaccharide is reduced only to the alditol obtained from the reduction of 1. D-talose? 2. D-glucose? 3. D-galactose?
b. What monosaccharide is reduced to two alditols, one of which is the alditol obtained from the reduction of 1. D-talose? 2. D-allose?
What sugar is the C-3 epimer of D-fructose?
Draw the mechanism for the dehydration step in the Wohl degradation
Draw the mechanism for the formation of β-lactose from a α- D-galactose and β- D-glucose in dil HCl.
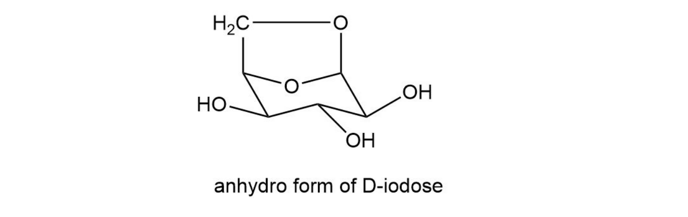
When a pyranose is in the chair conformation in which the CH2OH group and the C-1 OH group are both in axial positions, the two groups can react to form an acetal. This is called the anhydro form of the sugar (it has “lost water”). The anhydro form of d-idose is shown here. Explain why about 80% ofd-idose exists in the anhydro form in an aqueous solution at 100 °C, but only about 0.1% of d-glucose exists in the anhydro form under the same conditions.
What do you think about this solution?
We value your feedback to improve our textbook solutions.