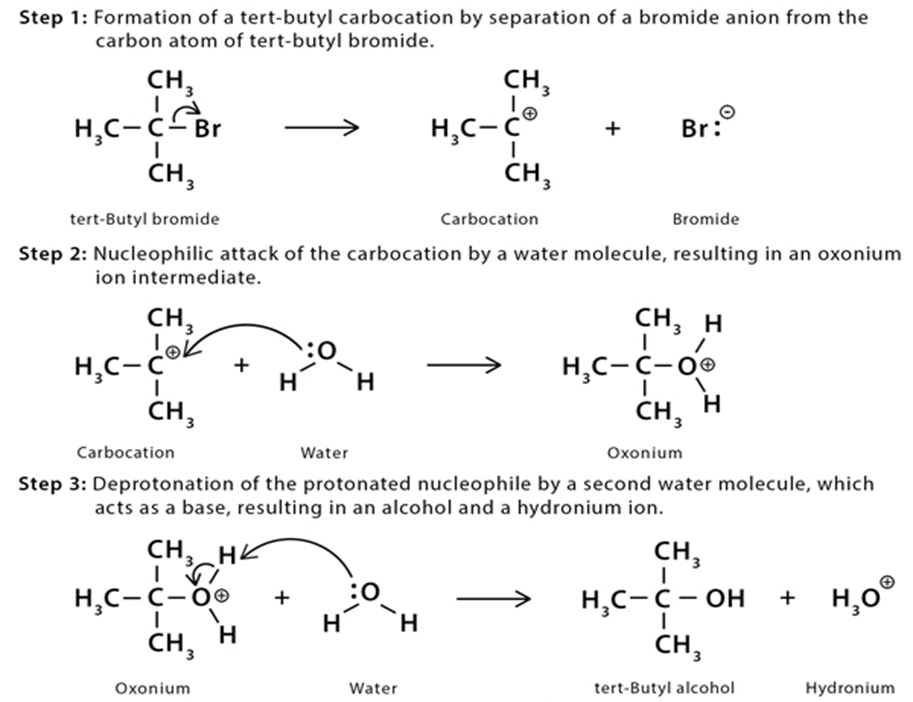
Chapter 9: Q114P (page 391)
Explain why the rate of the reaction of 1-bromo-2-butene with ethanol is increased if silver nitrate is added to the reaction mixture.
Short Answer
AgBr being an ionic compound, forms Ag+ in the solution which facilitates the formation of carbocation by forming AgBr after reaction. Thus, in presence of AgBr, the SN1 reaction facilitates.