Chapter 9: Q106P (page 391)
Show how each of the following compounds can be synthesized from the given starting materials:
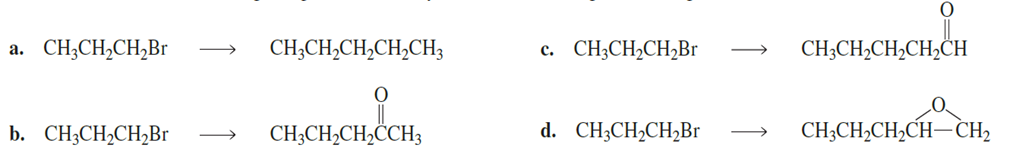
Short Answer
The mechanisms and the step-by-step processes of the reaction is given below,
Learning Materials
Features
Discover
Chapter 9: Q106P (page 391)
Show how each of the following compounds can be synthesized from the given starting materials:
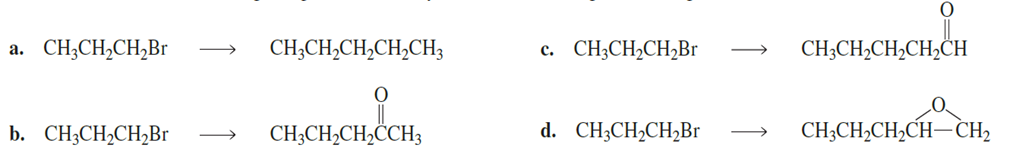
The mechanisms and the step-by-step processes of the reaction is given below,
All the tools & learning materials you need for study success - in one app.
Get started for free
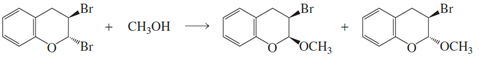
a. Propose a mechanism for the following reaction.
b. Explain why two products are formed.
c. Explain why methanol substitutes for only one of the bromines.
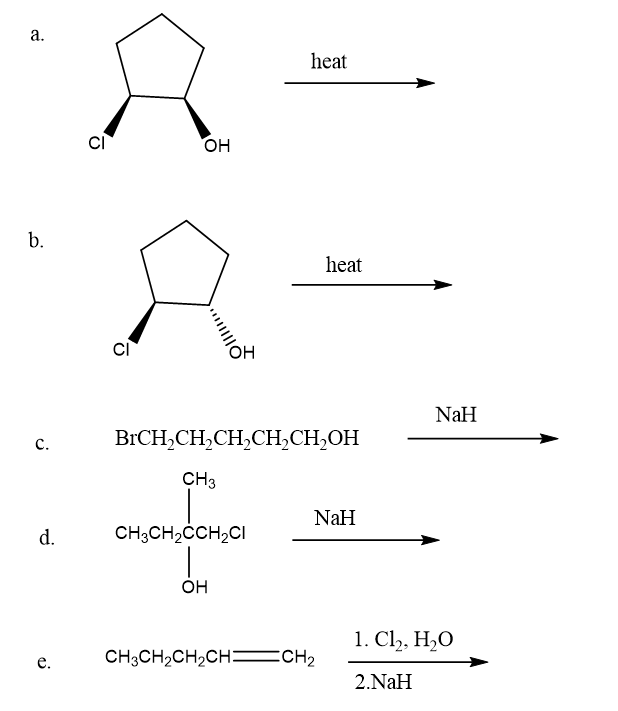
Draw the products of each of the following SN2/E2 reactions. If the products can exist as stereoisomers, show which stereoisomers are formed.
a.(3S,4S)-3-bromo-4-methylhexane + CH3O-
b.(3R,4R)-3-bromo-4-methylhexane + CH3O-
c.(3S,4R)-3-bromo-4-methylhexane + CH3O-
d.(3R,4S)-3-bromo-4-methylhexane + CH3O-
Draw the products of the following intramolecular reactions:
Starting with cyclohexene, how could the following compounds be prepared?
a. methoxycyclohexane
b. cyclohexylmethylamine
c. dicyclohexyl ether
Which species in each pair is more stable?
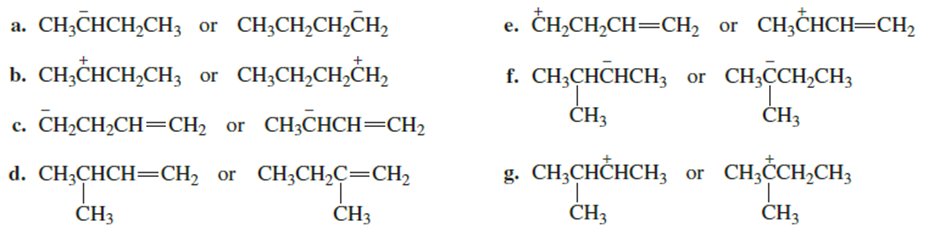
What do you think about this solution?
We value your feedback to improve our textbook solutions.