Chapter 1: Q4P (page 7)
How many valence electrons do the following atoms have?
- Boron 2. nitrogen 3. oxygen 4. Fluorine
Short Answer
Boron, nitrogen, oxygen, and fluorine have 3, 5, 6, and 7 valence electrons respectively.
Learning Materials
Features
Discover
Chapter 1: Q4P (page 7)
How many valence electrons do the following atoms have?
Boron, nitrogen, oxygen, and fluorine have 3, 5, 6, and 7 valence electrons respectively.
All the tools & learning materials you need for study success - in one app.
Get started for free
Imines can exist as stereoisomers. The isomers are named using the E, Z system of nomenclature (Section 4.2). The lone pair has the lowest priority.
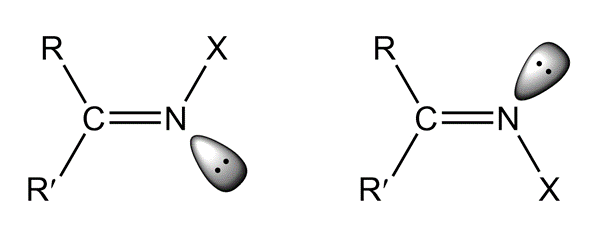
Draw the structure of each of the following compounds:
a. the (E)-hydrazone of benzaldehyde
b. the (Z)-oxime of propiophenone
Explain why the following compound is not stable
The compounds commonly known as “amino acids” are actually -aminocarboxylic acids (Section 21.0). What carbonyl compounds should be used to synthesize the two amino acids shown here?
Describe the orbital used in bonding and the bond angles in the following compounds:
a. CH3O-
b. CO2
c. H2CO
d. N2
e. BF3
Excess ammonia must be used when a primary amine is synthesized by reductive amination. What product will be obtained if the reaction is carried out with excess carbonyl compound?
What do you think about this solution?
We value your feedback to improve our textbook solutions.