Chapter 18: Q72P (page 918)
What products are obtained from the reaction of the following compounds with H2CrO4+ Δ?
a.
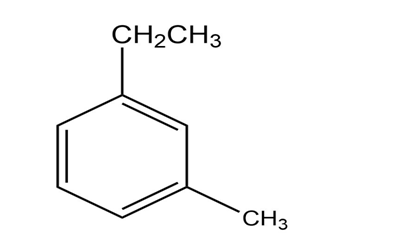
b.
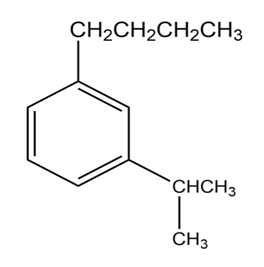
c.
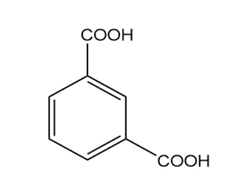
c. 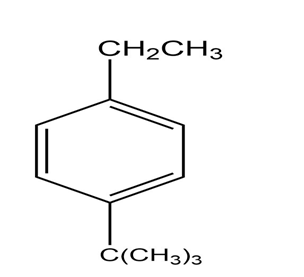
Short Answer
a.
b.
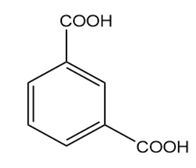
c.
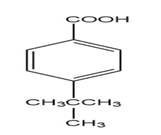
Learning Materials
Features
Discover
Chapter 18: Q72P (page 918)
What products are obtained from the reaction of the following compounds with H2CrO4+ Δ?
a.

b.

c.
c. 
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: The pKa values of a few ortho-, meta-, and para-substituted benzoic acids are shown below:
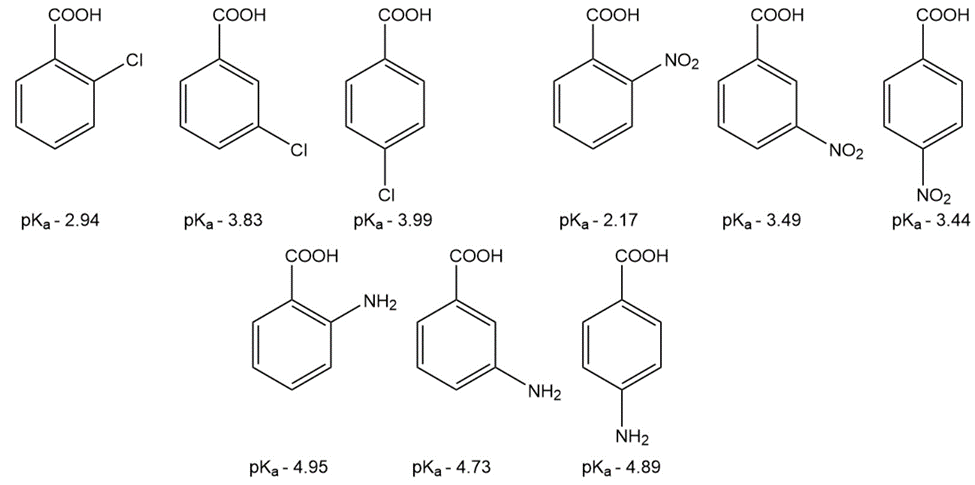
The relative pKa values depend on the substituent. For chloro-substituted benzoic acids, the ortho isomer is the most acidic and the para isomer is the least
acidic; for nitro-substituted benzoic acids, the ortho isomer is the most acidic and the meta isomer is the least acidic; and for amino-substituted benzoic acids, the meta isomer is the most acidic and the ortho isomer is the least acidic. Explain these relative acidities.
a. Cl: ortho > meta > para
b. NO2: ortho > para > meta
c. NH2: meta > para > ortho
What product is obtained from the reaction of excess benzene with each of the following reagents?
a. Ketoprofen, like ibuprofen, is an anti-inflammatory analgesic. How can ketoprofen be synthesised from the given starting material?
 Ketoprofen
Ketoprofen
b. ketoprofen and ibuprofen both have a propanoic acid substituent. Explain why the identical subunits are synthesised in different ways.
Draw the product of each of the following reactions:
a.
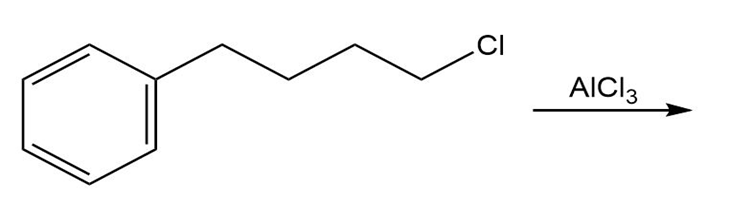
b.
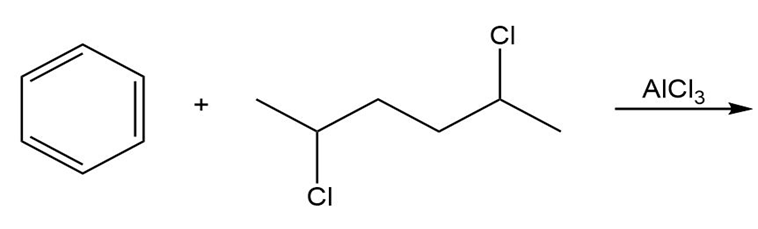
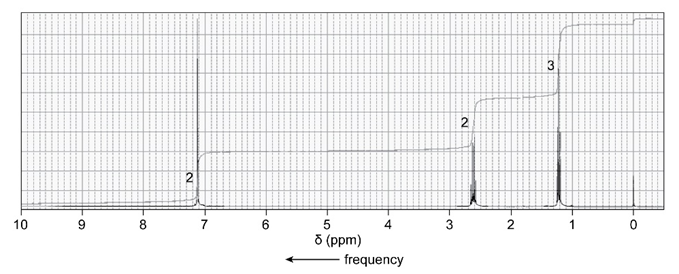
An unknown compound reacts with ethyl chloride and aluminum trichloride to form a compound that has the following 1H NMR spectrum. What is thestructure of the compound?
What do you think about this solution?
We value your feedback to improve our textbook solutions.