Chapter 10: Q8P (page 463)
What stereoisomers does the following reaction form?
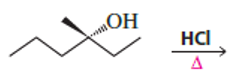
Short Answer
It will form two stereoisomers:
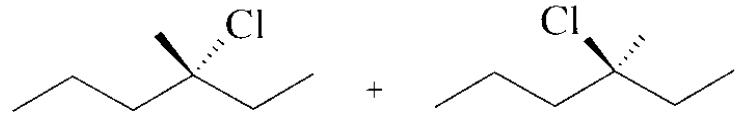
Learning Materials
Features
Discover
Chapter 10: Q8P (page 463)
What stereoisomers does the following reaction form?

It will form two stereoisomers:
All the tools & learning materials you need for study success - in one app.
Get started for free
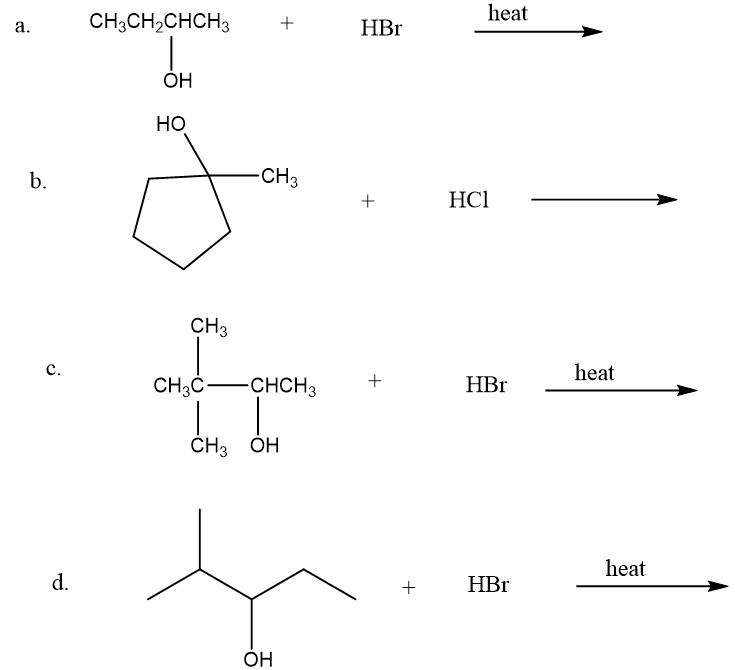
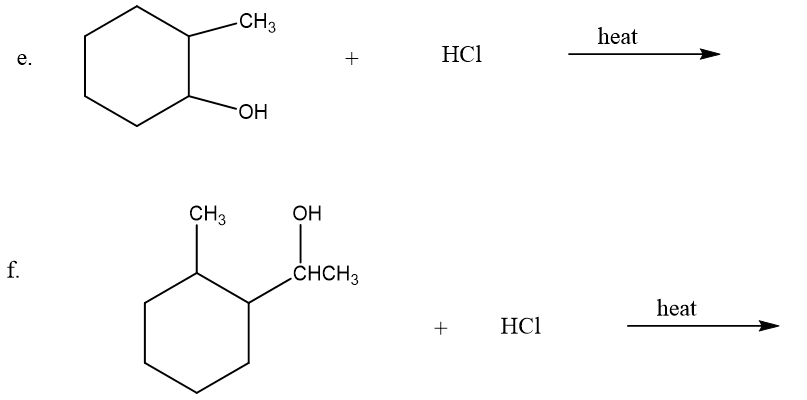
What is the major product of each of the following reactions?
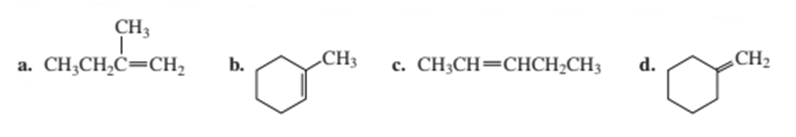
What alcohol would you treat with phosphorus oxychloride and pyridine to form each of the following alkenes?
Heating an alcohol with sulfuric acid is a good way to prepare a symmetrical ether such as diethyl ether.
a. Explain why it is not a good way to prepare an unsymmetrical ether such as ethyl propyl ether.
b. How would you synthesize ethyl propyl ether?
Why are NH3 and CH3NH2no longer nucleophiles when they are protonated?
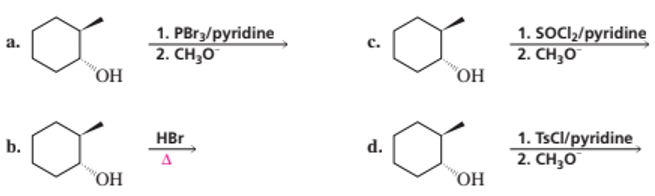
What stereoisomers do the following reactions form?
What do you think about this solution?
We value your feedback to improve our textbook solutions.