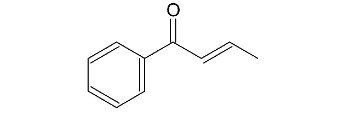
Chapter 17: Q88P (page 852)
Propose a mechanism for the following reaction.
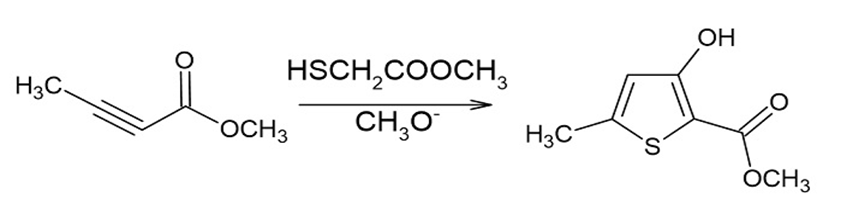
Short Answer
The proposed mechanism is shown below:
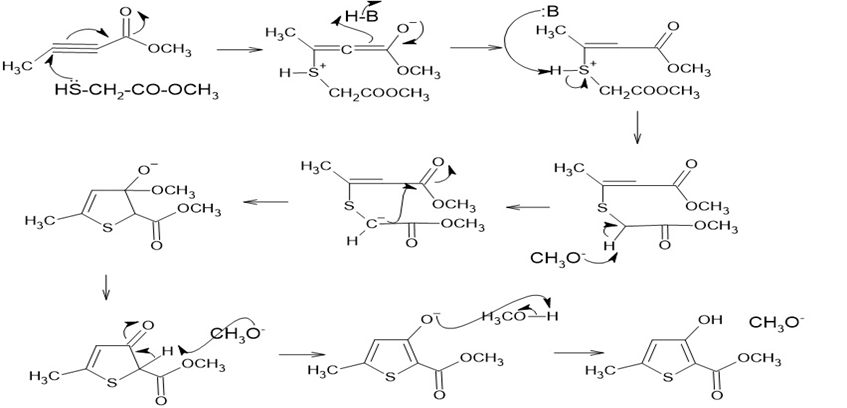
Learning Materials
Features
Discover
Chapter 17: Q88P (page 852)
Propose a mechanism for the following reaction.

The proposed mechanism is shown below:

All the tools & learning materials you need for study success - in one app.
Get started for free
Problem: starting with bromocyclohexane, how can each of the following compounds be prepared?
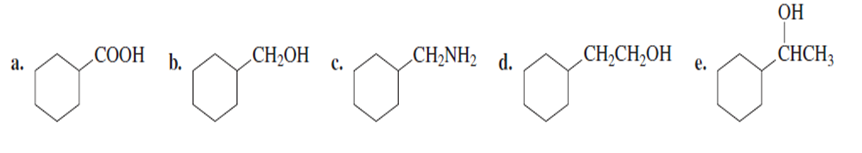
How could you prepare the following compound using a starting material that contains no more than three carbons?
Question: Explain why the -hydrogen of an N,N-disubstituted amide is less acidic (pKa= 30) than the hydrogen of an ester (pKa= 25).
The 1 H NMR chemical shifts of nitromethane, dinitromethane, and trinitromethane are at d 6.10, d 4.33, and d 7.52. Match each chemical shift with the compound. Explain how chemical shift correlates with pKa.
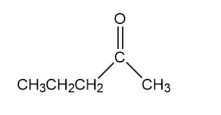
Describe three ways to synthesize the following compound:
What do you think about this solution?
We value your feedback to improve our textbook solutions.