Chapter 17: Q30P (page 826)
What is the product of each of the following reactions?
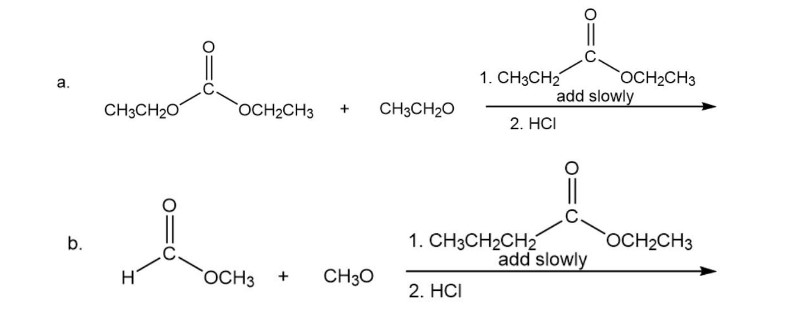
Short Answer
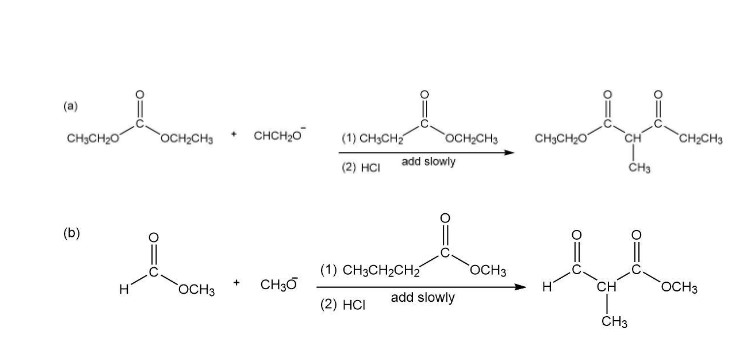
The product followed as:
Learning Materials
Features
Discover
Chapter 17: Q30P (page 826)
What is the product of each of the following reactions?

The product followed as:

All the tools & learning materials you need for study success - in one app.
Get started for free
What aldehyde or ketone would be obtained when each of the following compounds is heated in a basic aqueous solution?
(a) 2-ethyl-3-hydroxyhexanal
(b) 4-hydroxy-4-methyl-2-pentanone
(c) 2,4-dicyclohexyl-3-hydroxybutanal
(d) 5-ethyl-5-hydroxy-4-methyl-3-heptanone
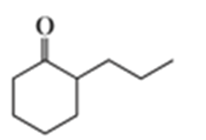
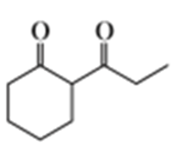
Describe how the following compounds could be prepared from cyclohexanone using an enamine intermediate:
a.
b.
A racemic mixture of 2-methyl-1-phenyl-1-butanone is formed when (R)-2-methyl-1-phenyl-1-butanone is dissolved in an acidic or basic aqueous solution. Give an example of another ketone that undergoes acid- or base-catalyzed racemization.
A student tried to prepare the following compounds using aldol condensations. Which of these compounds was she successful in synthesizing? Explain why the other syntheses were not successful.
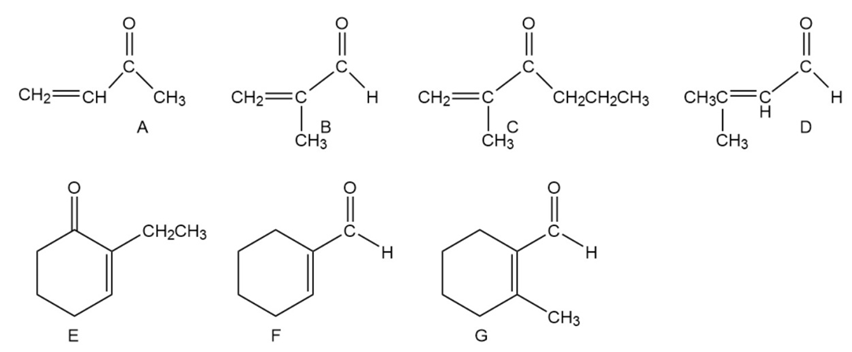
Indicate how each of the following compounds can be synthesized from the given starting material and any other necessary reagents:
What do you think about this solution?
We value your feedback to improve our textbook solutions.