Chapter 17: Q17P (page 815)
Describe how the following compounds could be prepared from cyclohexanone using an enamine intermediate:
a.
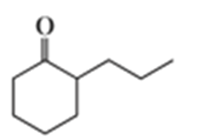
b.
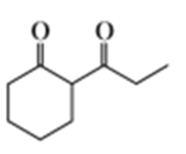
Short Answer
a.
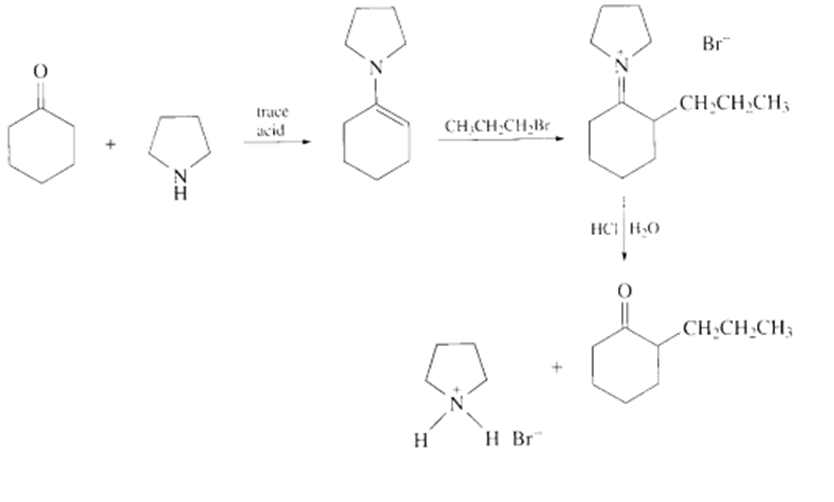
b.
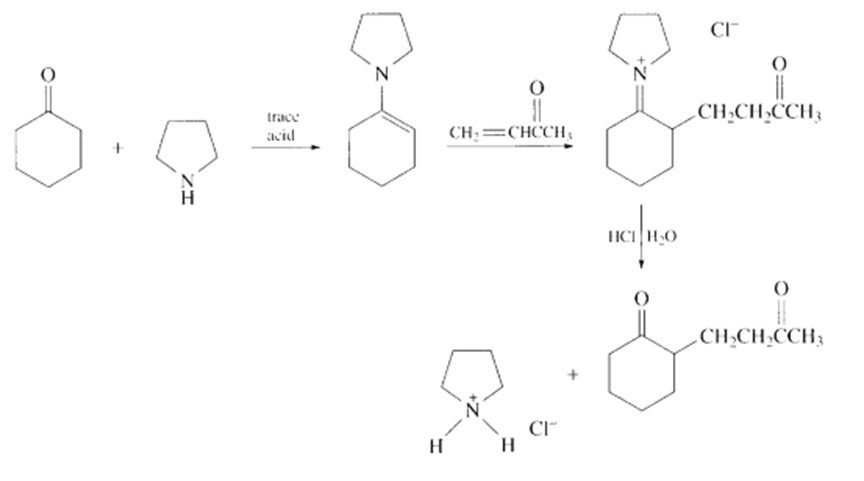
Learning Materials
Features
Discover
Chapter 17: Q17P (page 815)
Describe how the following compounds could be prepared from cyclohexanone using an enamine intermediate:
a.

b.

a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the products of the following reactions:
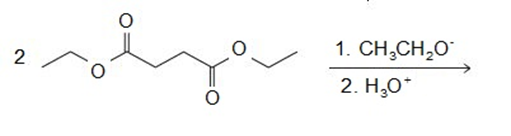
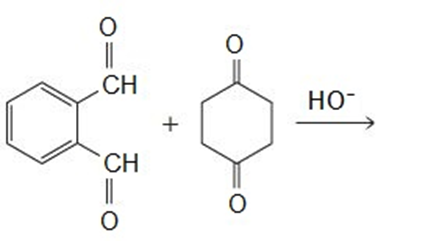
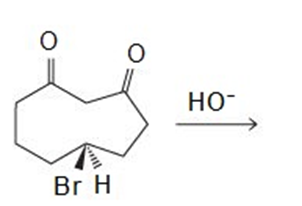
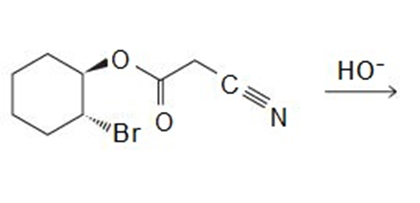
A -unsaturated carbonyl compound rearranges to a more stable conjugated -unsaturated compound in the presence of either acid or base.
a. Propose a mechanism for the base- catalyzed rearrangement.
b. Propose a mechanism for the acid- catalyzed rearrangement.
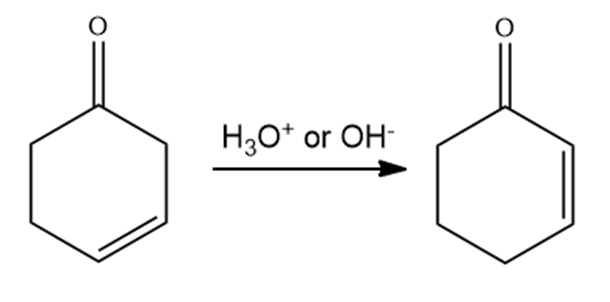
-unsaturated carbonyl compound -unsaturated carbonyl compound
There are other condensation reactions similar to the aldol and claisen condensations:
a. The Perkin condensation is the condensation of an aromatic aldehyde and acetic anhydride. Draw the product obtained from the following Perkin condensation:
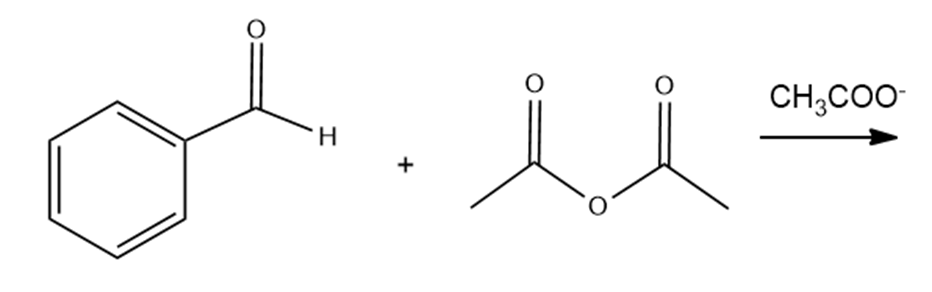
b. What compound is formed if water is added to the product of a Perkin condensation?
c. The Knoevenagel condensation is the condensation of an aldehyde or a ketone that has no alpha- hydrogens and a compound such as diethyl malonate that has an alpha-carbon flanked by two electron- withdrawing groups. Draw the product obtained from the following Knoevenagel condensation:
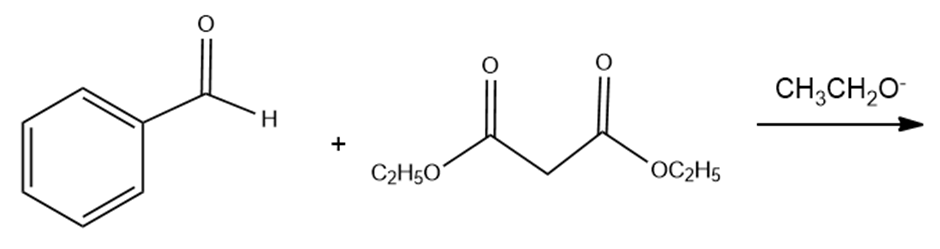
d. What product is obtained when the product of a Knoevenagel condensation is heated in an aqueous acidic solution?
Problem:What reagents are required to convert the reactant to the product?
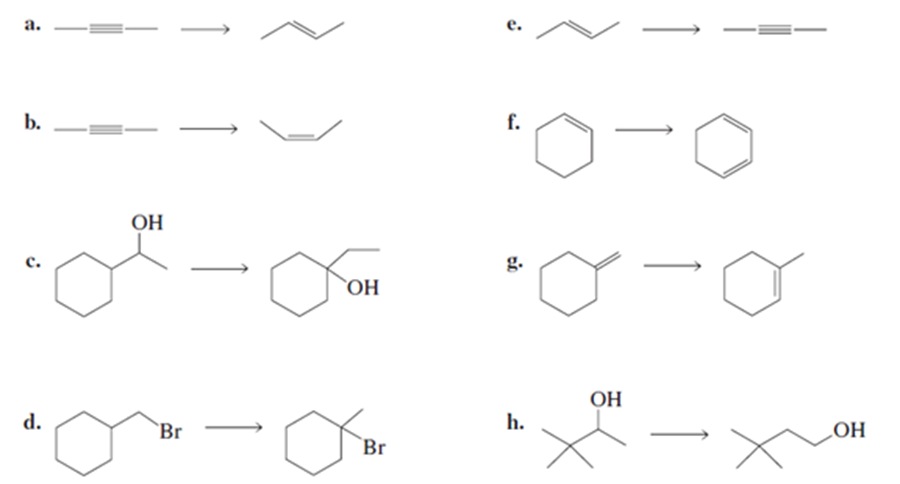
Show how 4-methyl-3-hexanol can be synthesised from 3-pentanone.
What do you think about this solution?
We value your feedback to improve our textbook solutions.