Chapter 17: Q 8 TP (page 859)
Do a retrosynthetic analysis on each of the following compounds, ending with available starting materials.
(a)
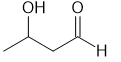
(b)
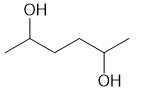
(C)
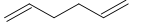
(D)
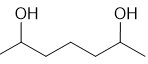
(e)
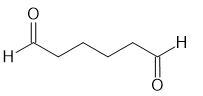
Short Answer
Answer:
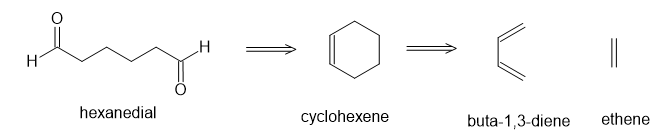
(a)
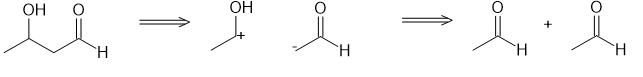
(b)
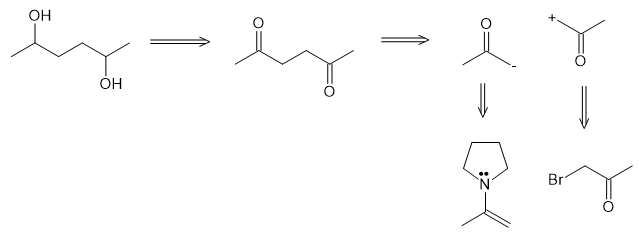
(c)
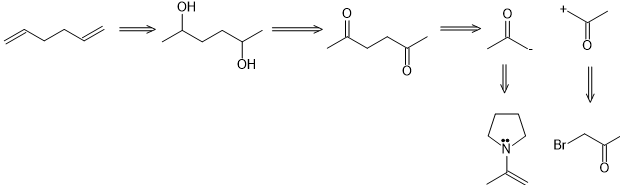
(d)
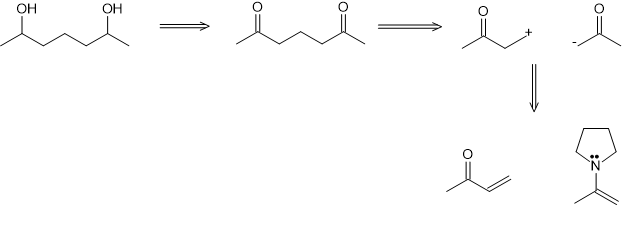
(e)
Learning Materials
Features
Discover
Chapter 17: Q 8 TP (page 859)
Do a retrosynthetic analysis on each of the following compounds, ending with available starting materials.
(a)

(b)

(C)

(D)

(e)

Answer:
(a)

(b)

(c)

(d)

(e)

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the products of the following reactions:
a. 1,3-cyclohexanedione + LDA/THF, followed by allyl bromide
b. g-butyrolactone + LDA/THF, followed by methyl iodide
c. 2,7-octanedione + sodium hydroxide
d. diethyl 1,2-benzenedicarboxylate + sodium ethoxide: (1) slow addition of ethyl acetate; (2) HCl
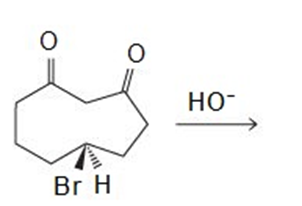
Draw the products of the following reactions:
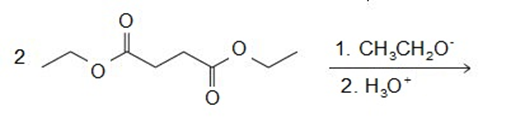
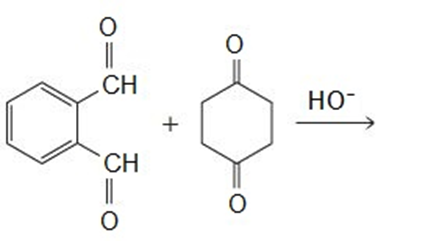
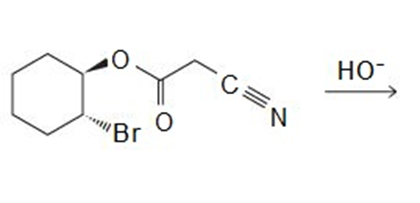
Explain why the following carboxylic acids cannot be prepared by a malonic ester synthesis:
Show how the following compounds can be synthesized from the given starting materials:
(a)
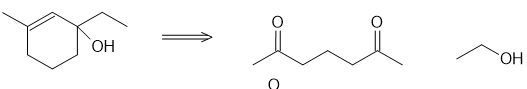
(b)
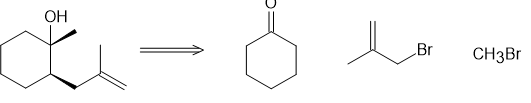
How could each of the following compounds be prepared from a ketone and an organohalide?
a.
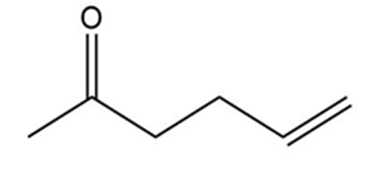
b.
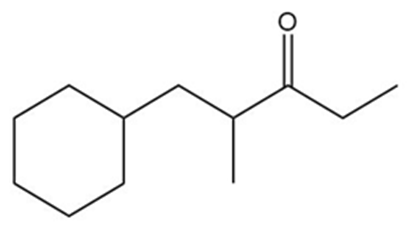
What do you think about this solution?
We value your feedback to improve our textbook solutions.