Chapter 17: 17-57P (page 847)
An aldol addition can be catalyzed by acids as well as by bases. Propose a mechanism for the acid-catalyzedaldol addition of propanal.
Short Answer
Learning Materials
Features
Discover
Chapter 17: 17-57P (page 847)
An aldol addition can be catalyzed by acids as well as by bases. Propose a mechanism for the acid-catalyzedaldol addition of propanal.

All the tools & learning materials you need for study success - in one app.
Get started for free
When the enzymatic decarboxylation of acetoacetate is carried out in, all the acetone that is formed contains. What does this tell you about the mechanism of the reaction?
Propose a mechanism for the following reaction:
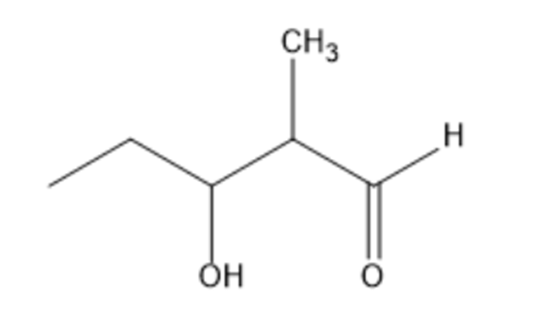
How could you prepare the following compound using a starting material that contains no more than three carbons?
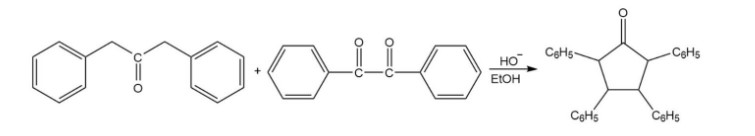
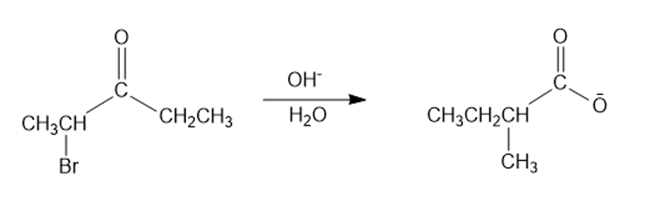
A carboxylic acid is formed when an -haloketone reacts with hydroxide ion. This reaction is called a Favorskii reaction. Propose a mechanism for the following Favorskii reaction.
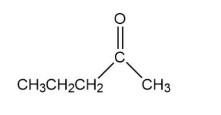
A -unsaturated carbonyl compound rearranges to a more stable conjugated -unsaturated compound in the presence of either acid or base.
a. Propose a mechanism for the base- catalyzed rearrangement.
b. Propose a mechanism for the acid- catalyzed rearrangement.
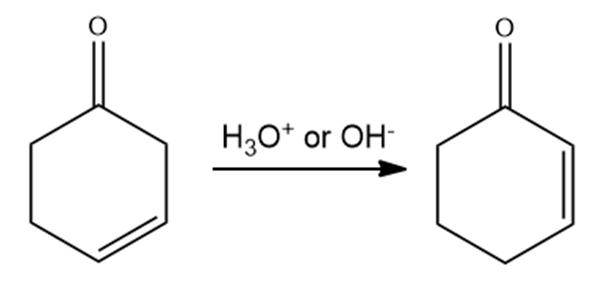
-unsaturated carbonyl compound -unsaturated carbonyl compound
What do you think about this solution?
We value your feedback to improve our textbook solutions.