Chapter 28: Q26P (page 1237)
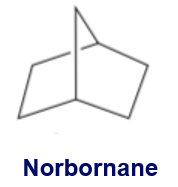
Show how norbornane can be prepared from cyclopentadiene.
Short Answer
Conversion ofcyclopentadiene to norbornane
Learning Materials
Features
Discover
Chapter 28: Q26P (page 1237)
Show how norbornane can be prepared from cyclopentadiene.

Conversion ofcyclopentadiene to norbornane

All the tools & learning materials you need for study success - in one app.
Get started for free
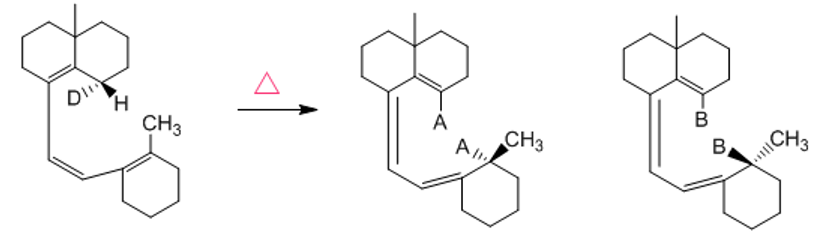
Two products are formed in the following [1,7] sigmatropic rearrangement, one due to hydrogen migration and the other to deuterium migration. Show the configuration of the products by replacing A and B with the appropriate atoms (H or D).
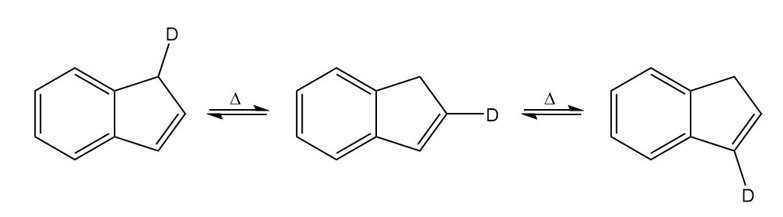
A student found that heating any one of the isomers shown here resulted in scrambling of the deuterium to all three positions on the five-membered ring. Propose a mechanism to account for this observation.
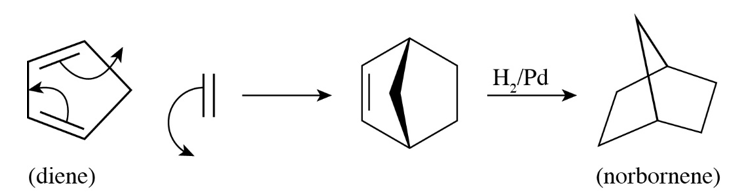
a. Propose a mechanism for the following reaction. (Hint:An electrocyclic reaction is followed by a Diels–Alder reaction.)
b. What would be the product if trans-2-butene were used instead of ethene?
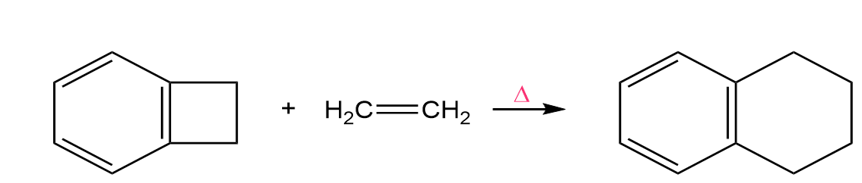
Problem:a. For conjugated systems with two, three, four, five, six, and seven conjugated bonds, construct quick MOs (just draw the lobes at the ends of the conjugated system as they are drawn on pages 1220 and 1221) to show whether the HOMO is symmetric or antisymmetric.
b. Using these drawings, convince yourself that the Woodward–Hoffmann rules in Table 28.1 are valid.
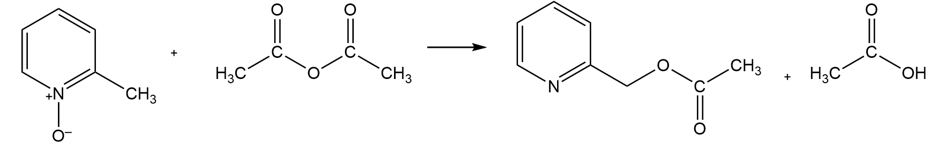
Propose a mechanism for the following reaction:
What do you think about this solution?
We value your feedback to improve our textbook solutions.