Chapter 14: Q62P (page 673)
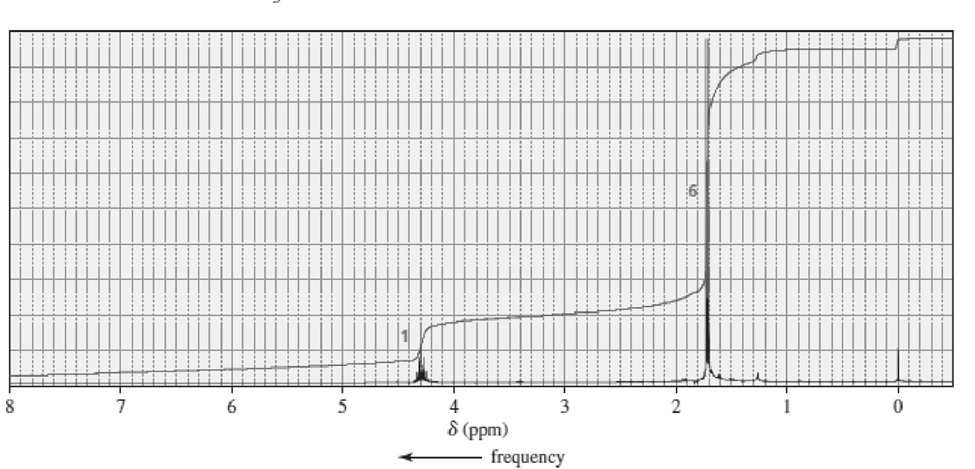
The 1H NMR spectra of three isomers with molecular formula C7H14O are shown here. Which isomer produces which spectrum?
a.

b.
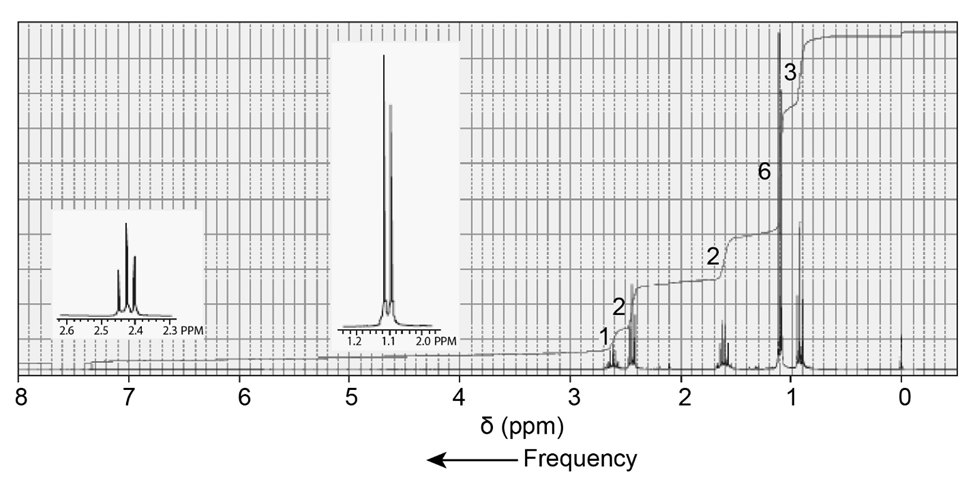
c.
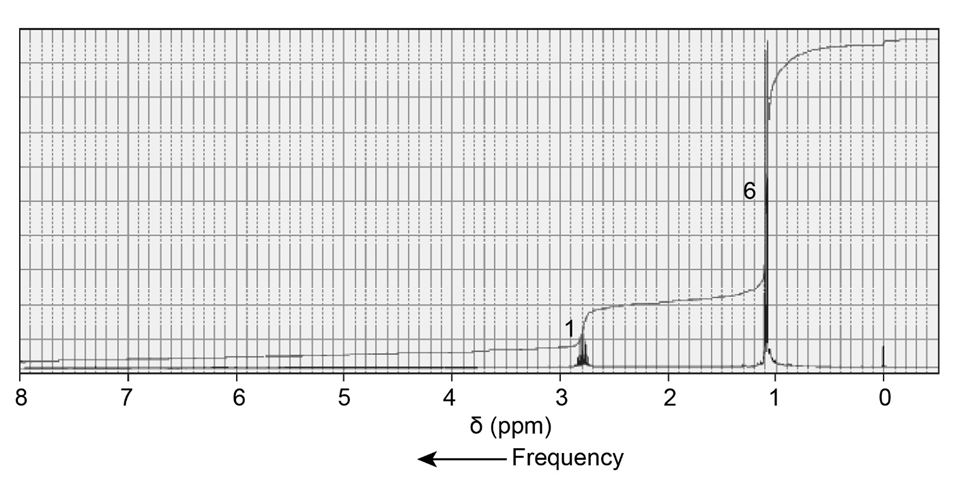
Short Answer
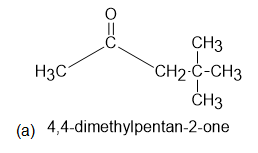
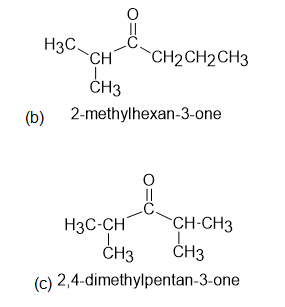
Learning Materials
Features
Discover
Chapter 14: Q62P (page 673)
The 1H NMR spectra of three isomers with molecular formula C7H14O are shown here. Which isomer produces which spectrum?
a.

b.

c.



All the tools & learning materials you need for study success - in one app.
Get started for free
Sketch the following spectra that would be obtained for 2-chloroethnol:
a. The 1H NMR spectrum for an anhydrous sample of the alcohol.
b. The 1H NMR spectrum for a sample of the alcohol that contains a trace amount of acid.
c. The 13C NMR spectrum.
d. The proton-coupled 13C NMR spectrum.
e. The four parts of a DEPT13C NMR spectrum.
Identify the compound with molecular formula C6H14 that is responsible for the following 1H NMR spectrum:

Match each of the 1HNMR spectra with one of the following compounds:
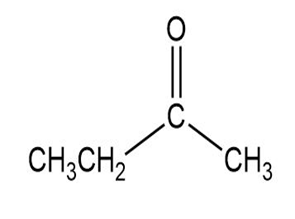
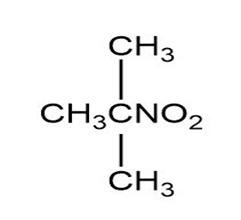

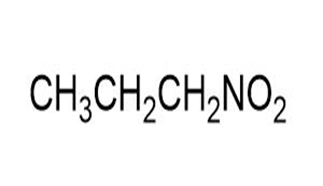
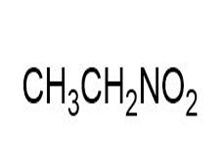

a.
b.
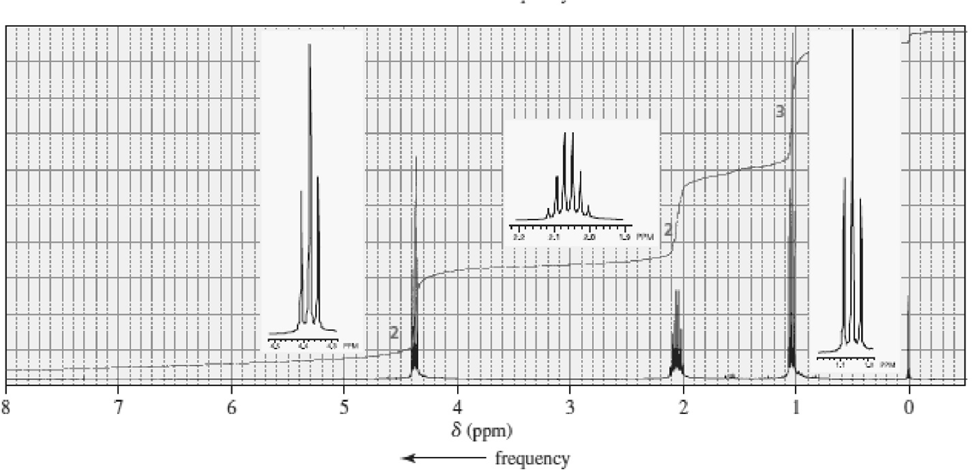
c.

Dr. N. M. Arr was called in to help analyze the 1 H NMR spectrum of a mixture of compounds known to contain only C, H, and Br. The mixture showed two singlets-one at 1.8 ppm and the other at 2.7 ppm-with relative integrals of 1: 6, respectively. Dr. Arr determined that the spectrum was that of a mixture of bromomethane and 2-Bromo-2-methylpropane. What was the ratio of bromomethane to 2-Bromo-2-methylpropane in the mixture?
An alkyl halide reacts with an alkoxide ion to form a compound whose 1H NMR spectrum is shown here. Identify the alkyl halide and the alkoxide ion.
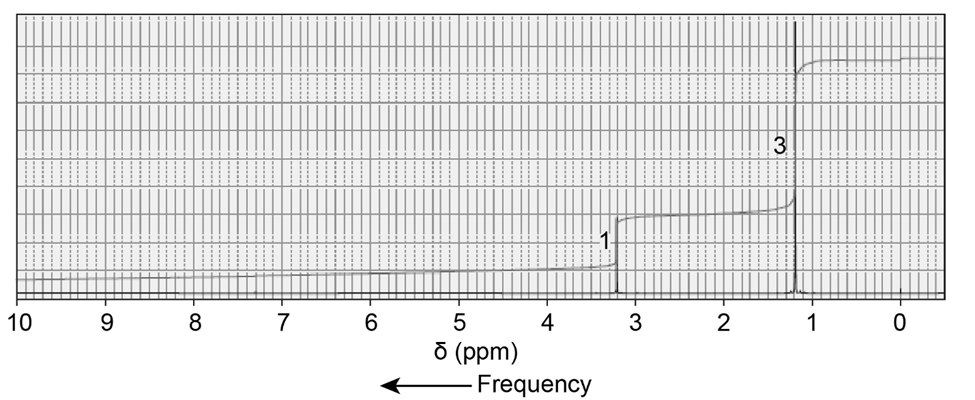
What do you think about this solution?
We value your feedback to improve our textbook solutions.