Chapter 14: Q43P (page 660)
How can 1,2-, 1,3-, and 1,4-dinitrobenzene be distinguished by
- 1H NMR spectroscopy?
- 13C NMR spectroscopy?
Short Answer
- 1,2-, 1,3-, and 1,4-dinitrobenzene can be distinguished by calculating their number of signals in 1H NMR spectroscopy.
- 1,2-, 1,3-, and 1,4-dinitrobenzene can be distinguished by observing the number of signals in 13C NMR spectroscopy.
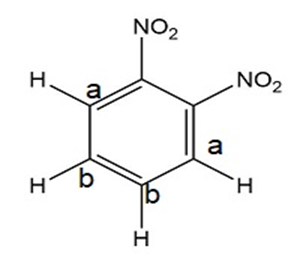
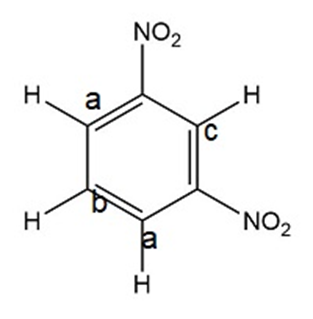
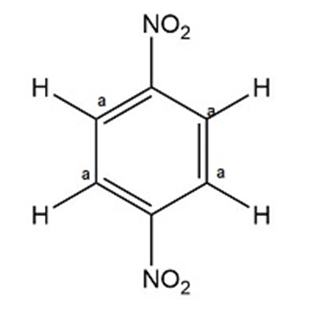
 Non-equivalent carbons
Non-equivalent carbons Non-equivalent carbons
Non-equivalent carbons Non-equivalent carbons
Non-equivalent carbons








