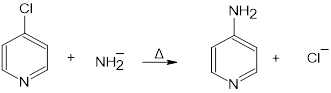
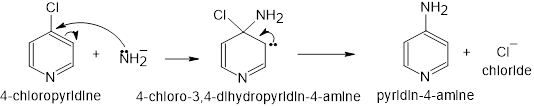
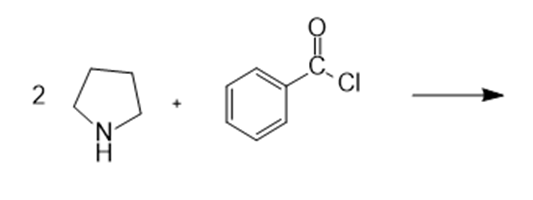
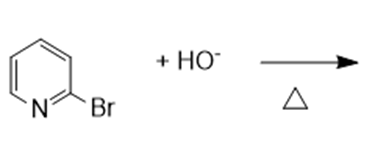
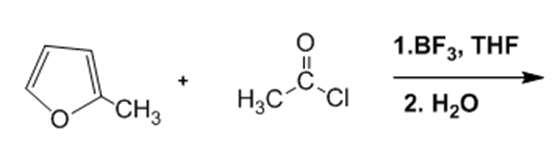
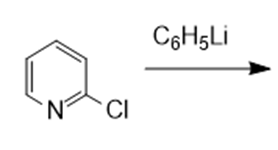
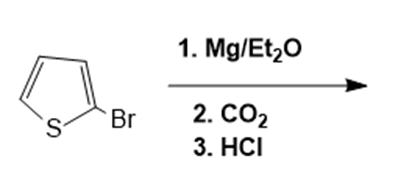
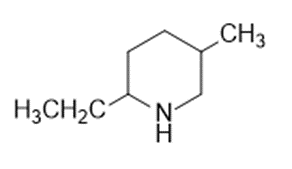
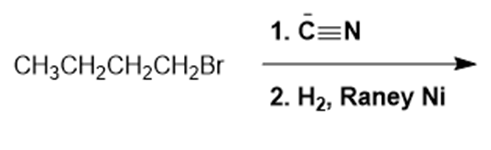In nucleophilic aromatic substitution reactions, the ring has a leaving group that is replaced by a nucleophile.Nucleophilic aromatic substitution of pyridine takes place at C-2 or C-4 because addition to these positions leads to the most stable intermediate. Only when addition occurs to these positions, a resonance contributor obtained that has the greatest electron density on nitrogen, the most electronegative elementof the ring atoms.
It involves two steps:
- The nucleophile added to the ring carbon that is attached to the leaving group.
- The leaving group is eliminated in the last step of the reaction.