Chapter 4: Q99P (page 185)
What is the configuration of each of the asymmetric centers in the following compounds?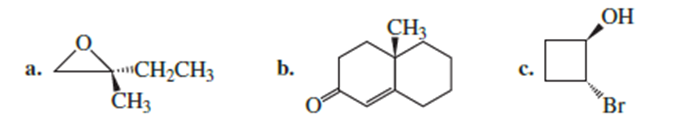
Short Answer
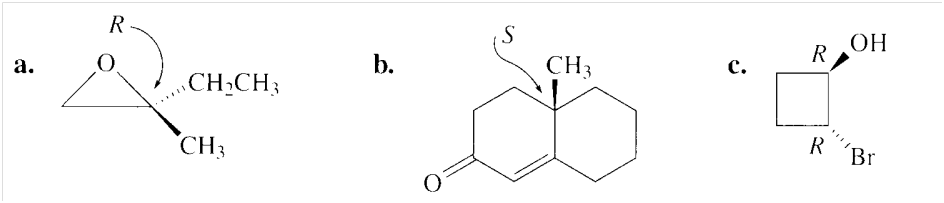
Here we have to find the configuration of each of the asymmetric centers in the following compounds:
Learning Materials
Features
Discover
Chapter 4: Q99P (page 185)
What is the configuration of each of the asymmetric centers in the following compounds?
Here we have to find the configuration of each of the asymmetric centers in the following compounds:

All the tools & learning materials you need for study success - in one app.
Get started for free
What is the configuration of each of the following?
a.
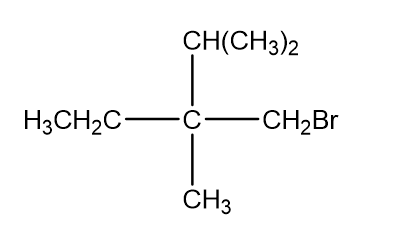
b.
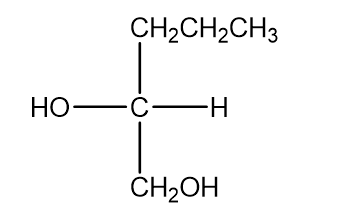
c.
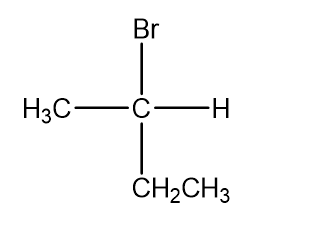
d.
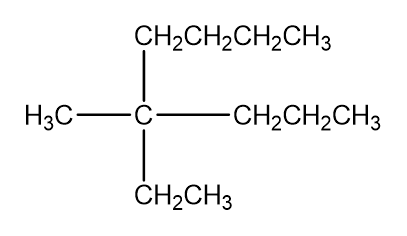
Draw four compounds with molecular formula C5H10that have carbon-carbon double bonds but do not have cis-trans isomers.
The stereoisomer of cholesterol found in nature is shown here.
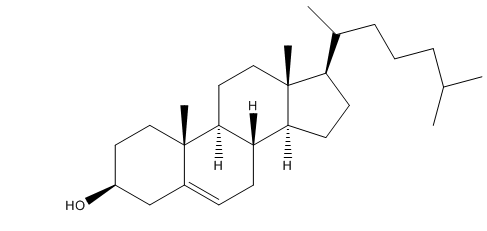
a. How many asymmetric centers does cholesterol have?
b. What is the maximum number of stereoisomers that cholesterol can have?
1-Bromo-2-methylcyclopentane has four pairs of diastereomers. Draw the four pairs
Assign relative priorities to each set of substituents:
a. -CH2CH2CH3 -CH(CH3)2 -CH=CH2 -CH3
b. -CH2NH2 -NH2 -OH -CH2OH
c. -C(=O)CH3 -CH=CH2 -Cl -C≡N
What do you think about this solution?
We value your feedback to improve our textbook solutions.