Chapter 4: Q40P (page 166)
The stereoisomer of cholesterol found in nature is shown here.
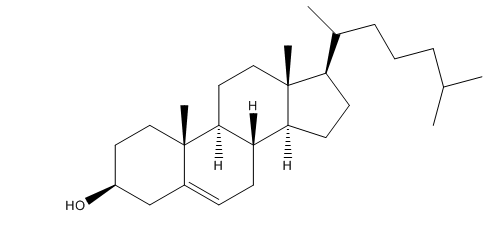
a. How many asymmetric centers does cholesterol have?
b. What is the maximum number of stereoisomers that cholesterol can have?
Short Answer
a. 8
b. 256
Learning Materials
Features
Discover
Chapter 4: Q40P (page 166)
The stereoisomer of cholesterol found in nature is shown here.

a. How many asymmetric centers does cholesterol have?
b. What is the maximum number of stereoisomers that cholesterol can have?
a. 8
b. 256
All the tools & learning materials you need for study success - in one app.
Get started for free
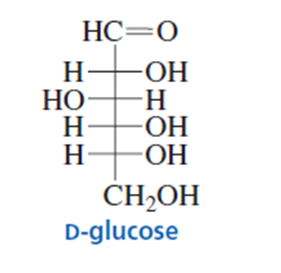
A student decided that the configuration of the asymmetric centers in a sugar such as d-glucose could be determined rapidly by simply assigning the Rconfiguration to an asymmetric center with an OH group on the right and the Sconfiguration to an asymmetric center with an OH group on the left. Is
he correct? (We will see in Chapter 20 that the “d” in d-glucose means that the OH group on the bottommost asymmetric center is on the right.)
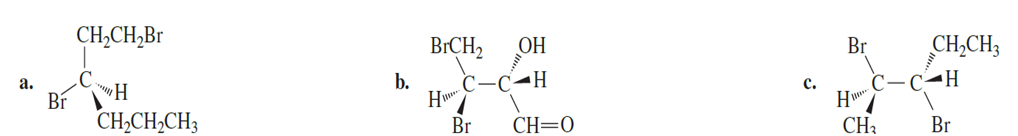
Question:What is the configuration of the asymmetric centers in the following compounds?
1-Bromo-2-methylcyclopentane has four pairs of diastereomers. Draw the four pairs
Question: Name the following compounds using R,S designations:
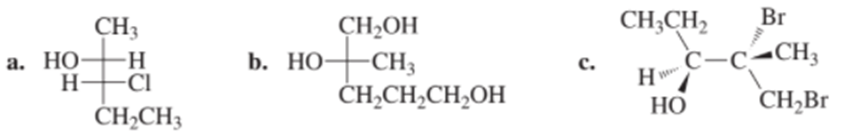
Disregarding stereoisomers, draw the structures of all alkenes with molecular formula C5H10. Which ones can exist as cis–trans isomers?
What do you think about this solution?
We value your feedback to improve our textbook solutions.