Chapter 4: Q11P (page 150)
Name each of the following:

Short Answer
The names of the given skeletal structures are:
a) (E)-2-heptene
b) (Z)-3,4-dimethyl-2-pentene
c) (Z)-1-chloro-3-ethyl-4-methyl-3-hexene
Learning Materials
Features
Discover
Chapter 4: Q11P (page 150)
Name each of the following:

The names of the given skeletal structures are:
a) (E)-2-heptene
b) (Z)-3,4-dimethyl-2-pentene
c) (Z)-1-chloro-3-ethyl-4-methyl-3-hexene
All the tools & learning materials you need for study success - in one app.
Get started for free
Name the following:
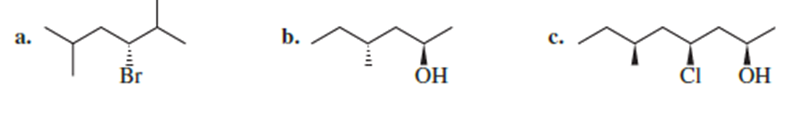
Which of the following has an asymmetric center?
CHBr2Cl BHFCl CH3CHCl2 CHFBrCl BeHCl
For each of the following structures, draw the most stable chair conformer.
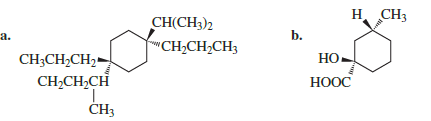
(+)-Mandelic acid has a specific rotation of +158. What would be the observed specific rotation of each of the following mixtures?
a. 50% (-)-mandelic acid and 50% (+)-mandelic acid
b. 25% (-)-mandelic acid and 75% (+)-mandelic acid
c. 75% (-)-mandelic acid and 25% (+)-mandelic acid
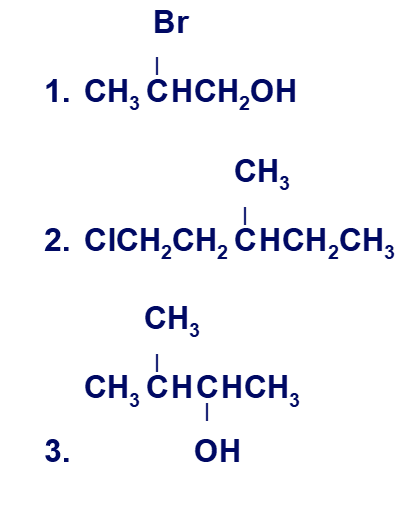
Draw enantiomers for each of the following using
a. perspective formulas.
b. Fischer's projections.
What do you think about this solution?
We value your feedback to improve our textbook solutions.