Chapter 4: Q-87P (page 184)
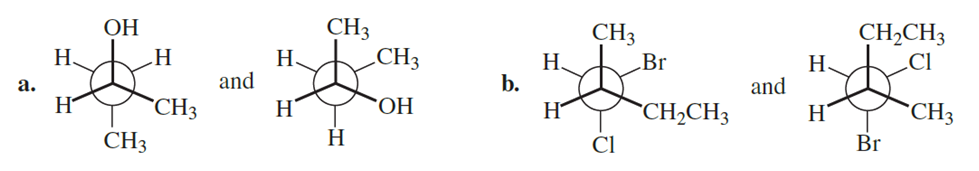
Question:Are the following pairs identical, enantiomers, diastereomers, or constitutional isomers?
Short Answer
a. The two structures in a are enantiomers.
b. The two structures in b are diastereomers.
Learning Materials
Features
Discover
Chapter 4: Q-87P (page 184)
Question:Are the following pairs identical, enantiomers, diastereomers, or constitutional isomers?

a. The two structures in a are enantiomers.
b. The two structures in b are diastereomers.
All the tools & learning materials you need for study success - in one app.
Get started for free
a.Which of the following compounds can exist as cis-trans isomers?
b. For those compounds that can exist as cis and trans isomers, draw and label the isomers.
1 CH3CH = CHCH2CH2CH3
2 CH3CH2 C = CHCH3
|
CH2CH3
3.CH3CH = CHCH3
4. CH3CH2CH=CH2
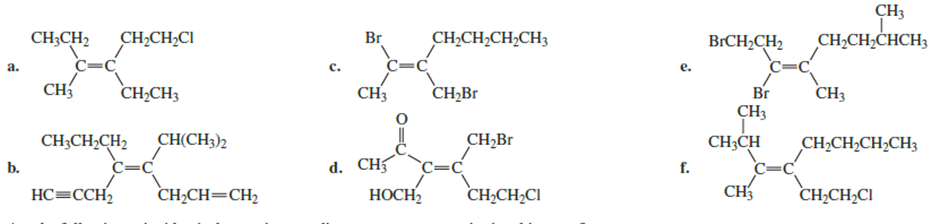
Do the following compounds have the E or the Z configuration?
1-Bromo-2-methylcyclopentane has four pairs of diastereomers. Draw the four pairs
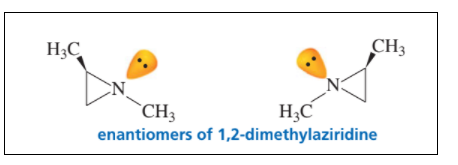
Explain why the enantiomers of 1,2-dimethylaziridine can be separated even though one of the “groups” attached to nitrogen is a lone pair.
Draw the cis and trans isomers for the following:
a. 1-bromo-4-chlorocyclohexane
b. 1-ethyl-3-methylcyclobutane
What do you think about this solution?
We value your feedback to improve our textbook solutions.