Chapter 8: Q3P (page 326)
a. Which of the following compounds have delocalized electrons?
b. Draw the resonance contributors for the compounds that have delocalized electrons.
1.CH2=CHCH2CH=CH2
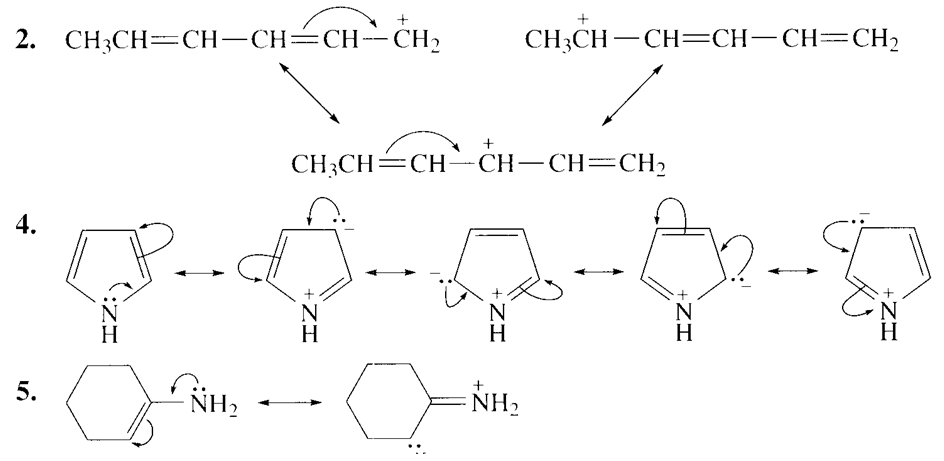
2.CH3CH=CHCH=CHCH2

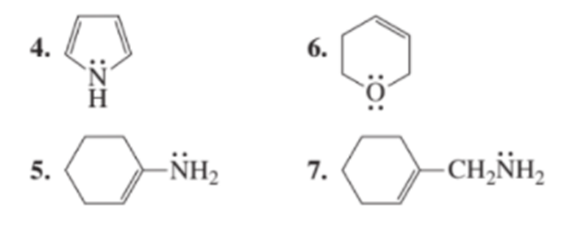
Short Answer
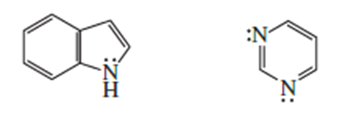
a) 2,4 and 5 have delocalized electrons.