Chapter 8: 8-97P (page 380)
What two sets of a conjugated diene and a dienophile could be used to prepare the following compound?
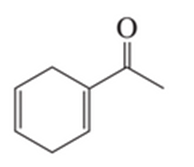
Short Answer
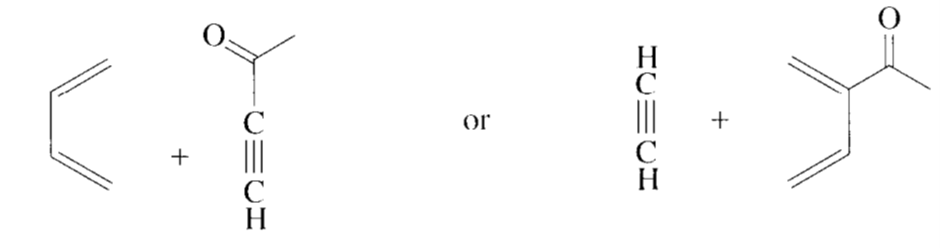
Learning Materials
Features
Discover
Chapter 8: 8-97P (page 380)
What two sets of a conjugated diene and a dienophile could be used to prepare the following compound?

All the tools & learning materials you need for study success - in one app.
Get started for free
On a single graph, draw the reaction coordinate diagram for the addition of one equivalent of HBr to 2 methyl-1,3-pentadiene and for the addition of one equivalent of HBr to 2-methyl-1,4-pentadiene. Which reaction is faster?
Draw the products obtained from the reaction of one equivalent of HBr with one equivalent of 1,3,5-hexatriene.
a. Which product(s) will predominate if the reaction is under kinetic control?
b. Which product(s) will predominate if the reaction is under thermodynamic control?
The heat of hydrogenation of 2,3-pentadiene, a cumulated diene, is 70.5 kcal/mol. What are the relative stabilities of cumulated, conjugated, and isolated dienes?
Draw all the products of the following reaction:
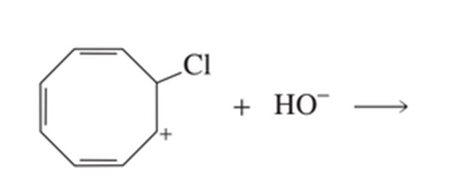
Which has the greater delocalization energy?
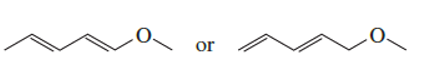
What do you think about this solution?
We value your feedback to improve our textbook solutions.