Chapter 3: Q60 P (page 137)
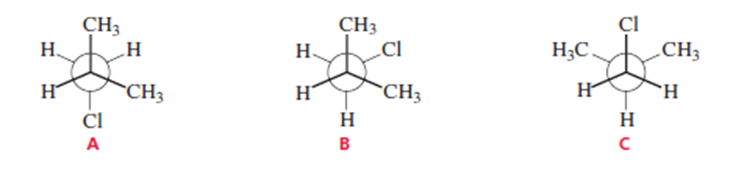
Which of the following conformers of isobutyl chloride is the most stable?
Short Answer
Conformer A is the most stable conformer of isobutyl chloride.
Learning Materials
Features
Discover
Chapter 3: Q60 P (page 137)
Which of the following conformers of isobutyl chloride is the most stable?

Conformer A is the most stable conformer of isobutyl chloride.
All the tools & learning materials you need for study success - in one app.
Get started for free
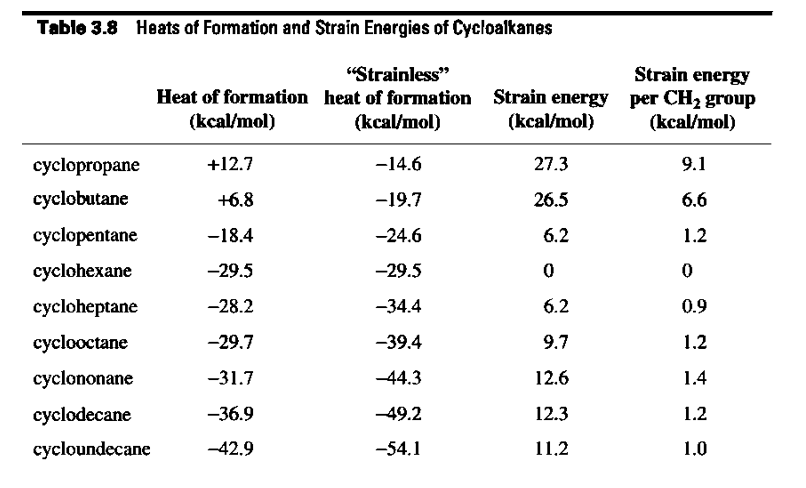
Verify the strain energy shown in Table 3.8 for cycloheptane.
What is each compound’s systematic name?

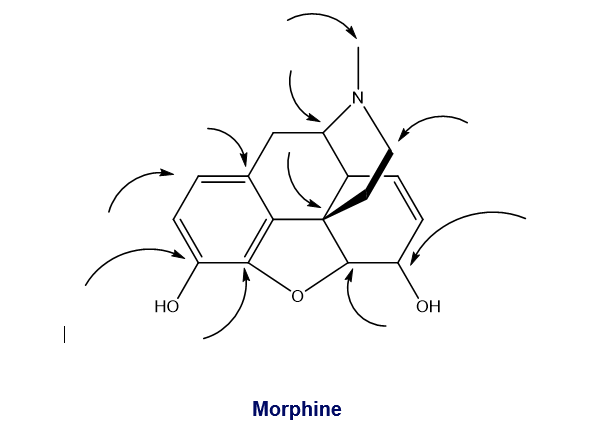
How many hydrogens are attached to each of the indicated carbons in morphine?
Explain why
a. H2O (100 °C) has a higher boiling point than CH3OH (65 °C)
b. H2O (100 °C) has a higher boiling point than NH3 (-33 °C).
c. H2O (100 °C) has a higher boiling point than HF (20 °C).
d. HF (20 °C) has a higher boiling point than NH3 (-33 °C).
Draw a condensed structure and a skeletal structure for each of the following:
a. sec-butyl tert-butyl ether
b. isoheptyl alcohol
c. sec-butylamine
d. isopentyl bromide
e. 5-(1-methylethyl)nonane
f. triethylamine
g. 4-(1,1-dimethylethyl)heptane
h. 5,5-dibromo-2-methyloctane
i. 3-ethoxy-2-methylhexane
j. 5-(1,2-dimethylpropyl)nonane
k. 3,4-dimethyloctane
l. 5-isopentyldecane
What do you think about this solution?
We value your feedback to improve our textbook solutions.