Chapter 21: Q40P (page 1018)
Explain why cyanogens bromide does not cleave on the C-side of cysteine.
Short Answer
The answer is
Imine is not formed due to the difficulty in the formation of the strained four-membered ring.
Learning Materials
Features
Discover
Chapter 21: Q40P (page 1018)
Explain why cyanogens bromide does not cleave on the C-side of cysteine.
The answer is
Imine is not formed due to the difficulty in the formation of the strained four-membered ring.
All the tools & learning materials you need for study success - in one app.
Get started for free
After the polypeptide shown below was treated with maleic anhydride, it was hydrolyzed by trypsin. (After a polypeptide is treated with maleic anhydride,
trypsin will cleave the polypeptide only on the C-side of arginine.)
Gly-Ala-Asp-Ala-Leu-Pro-Gly-Ile-Leu-Val-Arg-Asp-Val-Gly-Lys-Val-Glu-Val-Phe-Glu-Ala-Gly-
Arg-Ala-Glu-Phe-Lys-Glu-Pro-Arg-Leu-Val-Met-Lys-Val-Glu-Gly-Arg-Pro-Val-Gly-Ala-Gly-Leu-Trp
a. After a polypeptide is treated with maleic anhydride, why does trypsin no longer cleave it on the C-side of lysine?
b. How many fragments are obtained from the polypeptide?
c. In what order will the fragments be eluted from an anion-exchange column using a buffer of pH = 5?
An opioid pentapeptide has the following structure: Tyr-Cys-Gly-Phe-Cys
a. Draw the structure of the pentapeptide including all the side chains.
b. Write its structure following mild oxidation.
Cells can also convert a-keto acids into amino acids, but because the reagents organic chemists use for this reaction are not available in cells, they carry out this reaction by a different mechanism
(a) What amino acid is obtained from the reductive amination of each of the following metabolic intermediates in a cell by reductive amination?
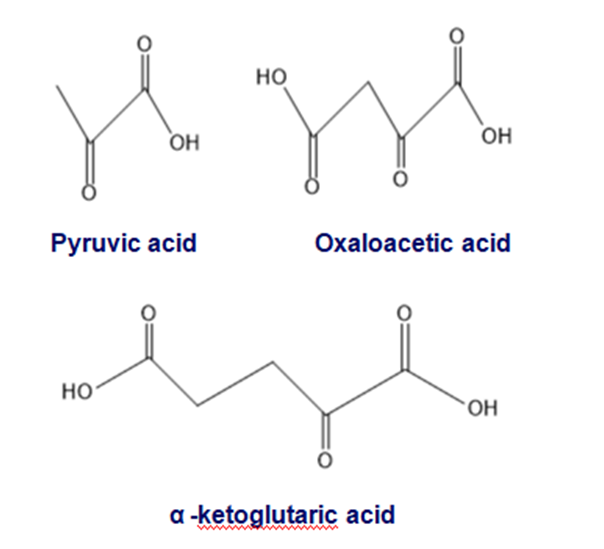
(b)What amino acids are obtained from the same metabolic intermediates when the amino acids are synthesized in the laboratory?
Which is the more effective buffer at physiological pH, a solution of 0.1 M glycylglycine or a solution of 0.2 M glycine?
Show the steps in the synthesis of the tetrapeptide in problem 34, using Merrifield’s method.
What do you think about this solution?
We value your feedback to improve our textbook solutions.