Chapter 21: Q30P (page 1007)
What is the configuration about each of the asymmetric centres in aspartame?
Short Answer
The answer is:
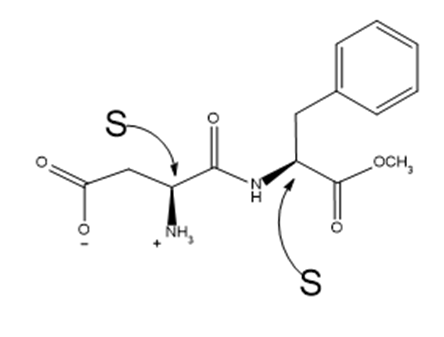
Configuration of Aspartame
Learning Materials
Features
Discover
Chapter 21: Q30P (page 1007)
What is the configuration about each of the asymmetric centres in aspartame?
The answer is:

Configuration of Aspartame
All the tools & learning materials you need for study success - in one app.
Get started for free
What dipeptides would be formed by heating a mixture of valine and N- protected leucine?
What amino acid is formed using the N-phthalimidomalonic ester synthesis when the following alkyl halides are used in the third step?
(a)
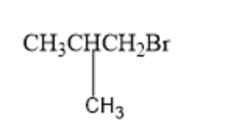
(b)
Why are the carboxylic acid groups of the amino acids more acidic
(pKa ~ 2) than a carboxylic acid such as acetic acid (pKa = 4.76)?
After the polypeptide shown below was treated with maleic anhydride, it was hydrolyzed by trypsin. (After a polypeptide is treated with maleic anhydride,
trypsin will cleave the polypeptide only on the C-side of arginine.)
Gly-Ala-Asp-Ala-Leu-Pro-Gly-Ile-Leu-Val-Arg-Asp-Val-Gly-Lys-Val-Glu-Val-Phe-Glu-Ala-Gly-
Arg-Ala-Glu-Phe-Lys-Glu-Pro-Arg-Leu-Val-Met-Lys-Val-Glu-Gly-Arg-Pro-Val-Gly-Ala-Gly-Leu-Trp
a. After a polypeptide is treated with maleic anhydride, why does trypsin no longer cleave it on the C-side of lysine?
b. How many fragments are obtained from the polypeptide?
c. In what order will the fragments be eluted from an anion-exchange column using a buffer of pH = 5?
A professor was preparing a manuscript for publication in which she reported that the pI of the tripeptide Lys-Lys-Lys was 10.6. One of her studentspointed out that there must be an error in her calculations because the pKa of the e-amino group of lysine is 10.8 and the pI of the tripeptide has to begreater than any of its individual pKa values. Was the student correct?
What do you think about this solution?
We value your feedback to improve our textbook solutions.