Chapter 2: Q6TP (page 83)
Which is the stronger acid?
a. CH3CH3 or HC ≡CH
b. H2C= CH2or HC≡ CH
c. H2C =CH2 or CH3CH3
Short Answer
a. HC ≡CH
b. HC≡ CH
c. H2C =CH2
Learning Materials
Features
Discover
Chapter 2: Q6TP (page 83)
Which is the stronger acid?
a. CH3CH3 or HC ≡CH
b. H2C= CH2or HC≡ CH
c. H2C =CH2 or CH3CH3
a. HC ≡CH
b. HC≡ CH
c. H2C =CH2
All the tools & learning materials you need for study success - in one app.
Get started for free
You are planning to carry out a reaction that produces protons. The reaction will be buffered at pH = 10.5. Would it be better to use a protonated methylamine/methylamine buffer or a protonated ethylamine/ethylamine buffer? (pKa of protonated methylamine = 10.7; pKaof protonated ethylamine = 11.0)
Rank the ions (-CH3, -NH2, HO-, and F-) from most basic to least basic.
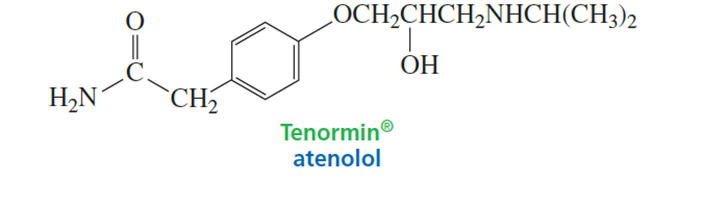
Tenormin, a member of the group of drugs known as beta-blockers, is used to treat high blood pressure and improve survival after a heart attack. It works by slowing down the heart to reduce its workload. Which atom in Tenormin is the most basic?
Which of the following are not acids?
CH3COOH CO2 HNO2 HCOOH CCl4
Question:Draw the products of the acid–base reaction when
a. HCl is the acid and NH3 is the base.
b. H2O is the acid and -NH2 is the base.
What do you think about this solution?
We value your feedback to improve our textbook solutions.