Chapter 2: Q20P (page 61)
Rank the ions (-CH3, -NH2, HO-, and F-) from most basic to least basic.
Short Answer
-CH3>-NH2,>HO->F-
Learning Materials
Features
Discover
Chapter 2: Q20P (page 61)
Rank the ions (-CH3, -NH2, HO-, and F-) from most basic to least basic.
-CH3>-NH2,>HO->F-
All the tools & learning materials you need for study success - in one app.
Get started for free
After examining the potential maps for LiH, HF and answer the following question:
Which compound has the hydrogen that would be most apt to attract a negatively charged molecule?
Question:Draw the products of the acid–base reaction when
a. HCl is the acid and NH3 is the base.
b. H2O is the acid and -NH2 is the base.
Calculate the equilibrium constant for the acid-base reaction between the reactants in each of the following pairs:
a.
b.
c.
d.
Consider the following reaction:
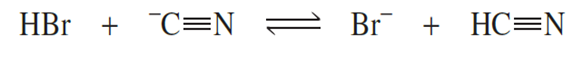
a. What is the acid on the left side of the equation?
b. What is the base on the left side of the equation?
c. What is the conjugate base of the acid on the left?
d. What is the conjugate acid of the base on the left?
e. What is the acid on the right side of the equation?
f. What is the base on the right side of the equation?
g. What is the conjugate base of the acid on the right?
h. What is the conjugate acid of the base on the right?
Question:An acid has a Ka of 4.53 ×10-6 in water. What is its Keq for reaction with water in a dilute solution?
([H2O] = 55.5 M)
What do you think about this solution?
We value your feedback to improve our textbook solutions.