Chapter 2: Q11P (page 57)
a. Write an equation showing CH3OH reacting with and an equation showing reacting with HCl.
b. Write an equation showing reacting as an acid with and an equation showing it reacting as a base with HBr.
Short Answer
a.
b.
Learning Materials
Features
Discover
Chapter 2: Q11P (page 57)
a. Write an equation showing CH3OH reacting with and an equation showing reacting with HCl.
b. Write an equation showing reacting as an acid with and an equation showing it reacting as a base with HBr.
a.
b.
All the tools & learning materials you need for study success - in one app.
Get started for free
Which of the following are not acids?
CH3COOH CO2 HNO2 HCOOH CCl4
Calculate the equilibrium constant for the acid-base reaction between the reactants in each of the following pairs:
a.
b.
c.
d.
a. For each of the acid-base reactions in section 2.3, compare the pKa values of the acids on either side of the equilibrium arrows to prove that the equilibrium lies in the direction indicated. (The pKa values you need are found in section 2.3 or in problem 13)
b. Do the same for the acid-base reaction in section 2.1
Consider the following reaction:
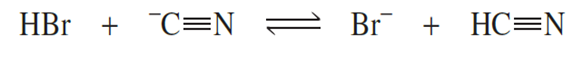
a. What is the acid on the left side of the equation?
b. What is the base on the left side of the equation?
c. What is the conjugate base of the acid on the left?
d. What is the conjugate acid of the base on the left?
e. What is the acid on the right side of the equation?
f. What is the base on the right side of the equation?
g. What is the conjugate base of the acid on the right?
h. What is the conjugate acid of the base on the right?
Butyric acid, the compound responsible for the unpleasant odor and taste of sour milk, has a pKa value of 4.82. What is its Ka value? Is it a stronger acid or a weaker acid than vitamin C?
What do you think about this solution?
We value your feedback to improve our textbook solutions.