Chapter 2: Q10P (page 57)
Question: Draw the conjugate acid for each of the following:
Learning Materials
Features
Discover
Chapter 2: Q10P (page 57)
Question: Draw the conjugate acid for each of the following:
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:An acid has a Ka of 4.53 ×10-6 in water. What is its Keq for reaction with water in a dilute solution?
([H2O] = 55.5 M)
Which is a stronger acid?
CH3OH+CH3 or 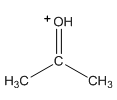
Which is the stronger acid?
a. CH3OH or CH3CH3
b. CH3OH or HF
c. CH3NH2 or HF
d. CH3NH2 or CH3OH
You are planning to carry out a reaction that produces protons. The reaction will be buffered at pH = 10.5. Would it be better to use a protonated methylamine/methylamine buffer or a protonated ethylamine/ethylamine buffer? (pKa of protonated methylamine = 10.7; pKaof protonated ethylamine = 11.0)
An atom with a formal charge does not necessarily have more or less electron density than the atoms in the molecule without formal charges. We can see this by examining the potential maps for and .
Which atom bears the formal negative charge in the hydroxide ion?
Which atom has greater electron density in the hydroxide ion?
Which atom bears the formal positive charge in the hydronium ion?
Which atom has the least electron density in the hydronium ion?
What do you think about this solution?
We value your feedback to improve our textbook solutions.