Chapter 4: The study of chemical reactions
Q 50P
(a) Draw the structure of the transition state for the second propagation step in the chlorination of methane.
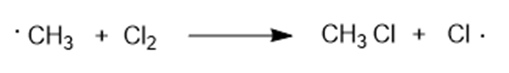
Show whether the transition state is product-like or reactant-like and which of the two partial bonds is stronger.
(b) Repeat for the second propagation step in the bromination of methane.
Q 51P
Peroxides are often added to free-radical reactions as initiators because the oxygen-oxygen bond cleaves homolytically rather easily. For example, the bond-dissociation enthalpy of O-Obond in hydrogen peroxide (H-O-O-H)is only 213 kJ/mol (51 kcal/mol). Give a mechanism for the hydrogen peroxide- initiated reaction of cyclopentane with chlorine. The BDE for HO-Clis 210 kJ/mol (50kcal/mol).
Q 52P
When dichloromethane is treated with strong NaOH , an intermediate is generated that reacts like a carbene. Draw the structure of this reactive intermediate, and propose a mechanism for its formation.
Q5P
The following reaction has a value of
(a) Calculate at room temperature for this reaction as written.
(b)Starting with a solution of and , calculate the final concentrations of all four species at equilibrium.
Q6P
Question: Under base-catalyzed conditions, two molecules of acetone can condense to form diacetone alcohol. At room temperature, aboutof 5%the acetone is converted to diacetone alcohol. Determine the value offor this reaction.
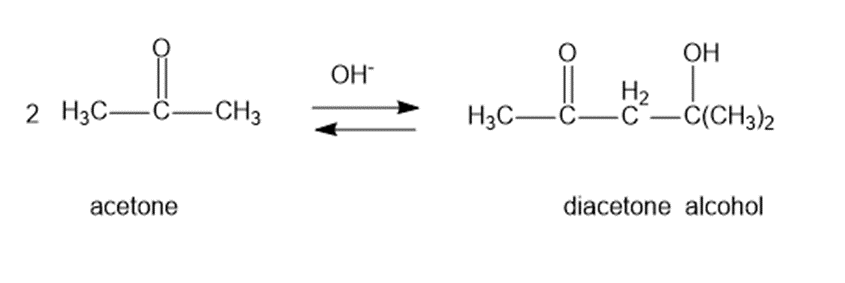
Q7P
Question: When ethene is mixed with hydrogen in the presence of a platinum catalyst, hydrogen adds across the double bond to form ethane. At room temperature, the reaction goes to completion. Predict the signs for this reaction. Explain these signs in terms of bonding and freedom of motion.
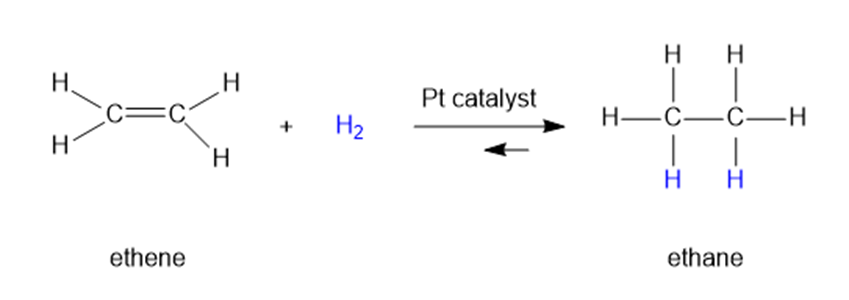
Q8P
Question: For each reaction, estimate whether for the reaction is positive, negative or impossible to predict.
a.
b.The formation of diacetone alcohol:
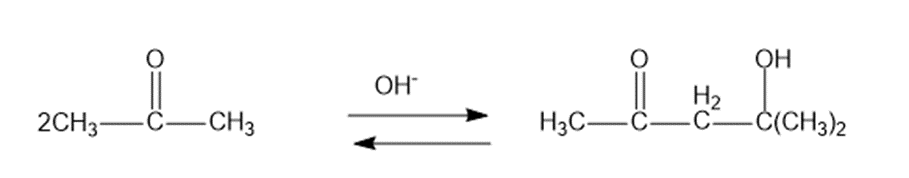
c.
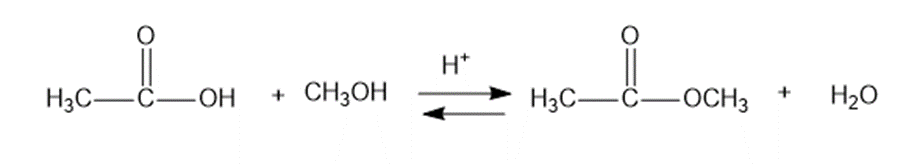
Q9P
Question: (a) Propose a mechanism for the free-radical chlorination of ethane.
(b) Calculatefor each step in this reaction
(c) Calculate the overall value offor this reaction.
