Chapter 7: Q57Q (page 332)
Question:
Show how you would prepare cyclohexene from each compound.
(a) cyclohexanol
(b) cyclohexyl bromide
(c) cyclohexane (not by dehydrogenation)
Short Answer


Learning Materials
Features
Discover
Chapter 7: Q57Q (page 332)
Question:
Show how you would prepare cyclohexene from each compound.
(a) cyclohexanol
(b) cyclohexyl bromide
(c) cyclohexane (not by dehydrogenation)


All the tools & learning materials you need for study success - in one app.
Get started for free
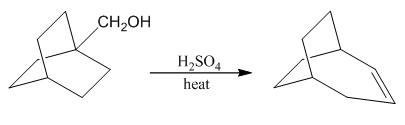
Write a mechanism that explains the formation of the following product. In your mechanism, explain the cause of the rearrangement, and explain the failure to form the Zaitsev product.
Question: Draw five more compounds of formula.
Show that the (S,S) enantiomer of this (R,R) diastereomer of 1-bromo-1,2-diphenylpropane also undergoes E2 elimination to give the cis diastereomer of the product. (We do not expect these achiral reagents to distinguish between enantiomers.)
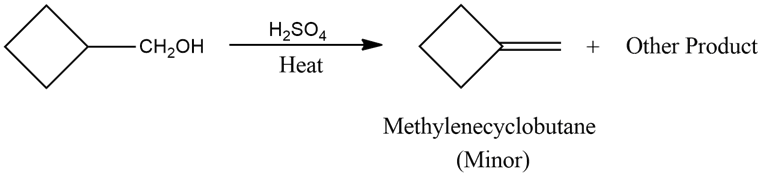
A graduate student wanted to make methylenecyclobutane, and he tried the following reaction. Propose structures for the other products, and give mechanisms to account for their formation.
Predict the elimination products of the following reactions and label the major products.
What do you think about this solution?
We value your feedback to improve our textbook solutions.