Chapter 10: Q31P (page 531)
Give systematic (IUPAC) names for the following diols and phenols.
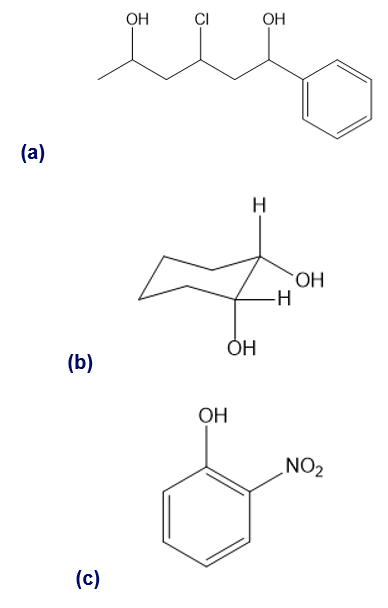
Short Answer
(a) 3-chloro-1-phenylhexane-1,5-diol
(b) cis-cyclohexane-1,2-diol
(c) 2-nitrophenol
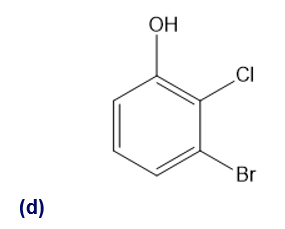
(d) 3-bromo-2-chlorophenol
Learning Materials
Features
Discover
Chapter 10: Q31P (page 531)
Give systematic (IUPAC) names for the following diols and phenols.


(a) 3-chloro-1-phenylhexane-1,5-diol
(b) cis-cyclohexane-1,2-diol
(c) 2-nitrophenol
(d) 3-bromo-2-chlorophenol
All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would synthesize the following:
Show how you would synthesize the following compounds from any starting materials containing no more than six carbon atoms.
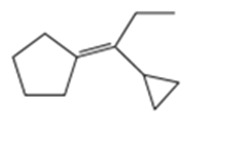
a)
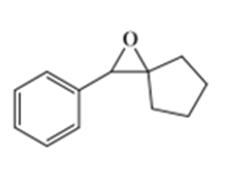
b)
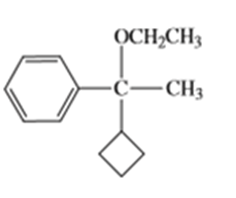
c)
Show how you would synthesize each tertiary alcohol by adding an appropriate Grignard reagent to a ketone.
(a)3-phenylhexane-3-ol (3ways)
(b) Ph3 COH
(c) 1-ethylcyclopentanol
(d) 2-cyclopentylpentan-2-ol
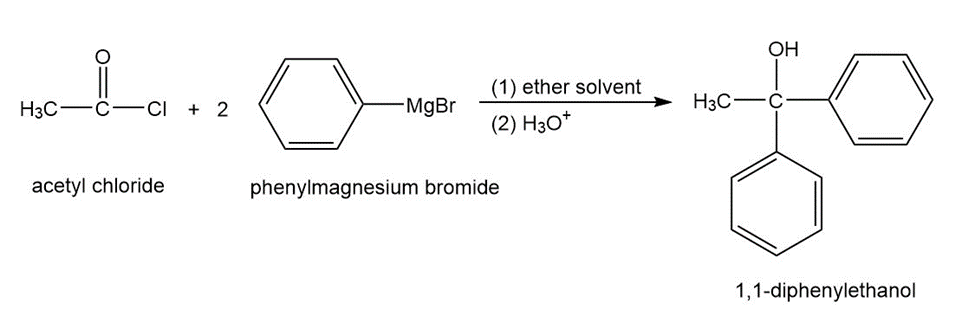
Propose a mechanism for the reaction of acetyl chloride with phenylmagnesium bromide to give 1,1-diphenylethanol.
Often, compounds can be synthesized by more than one method. Show how this 3° alcohol can be made from the following:
(a) two different ketones
(b) two different alkenes
(c) an ester
(d) a 3° alkyl bromide
What do you think about this solution?
We value your feedback to improve our textbook solutions.