Chapter 18: PROBLEM 18.34 (page 958)
Question: Predict the major products of the following reactions.
a.

b.
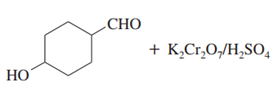
c.
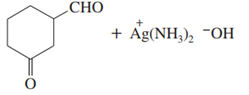
d.,
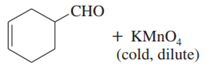
Short Answer
Answer
a.
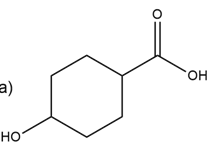
b.

c,
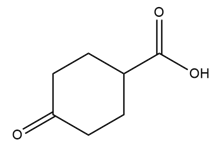
d.
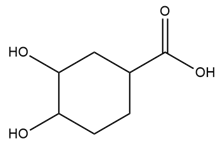
Learning Materials
Features
Discover
Chapter 18: PROBLEM 18.34 (page 958)
Question: Predict the major products of the following reactions.
a.

b.
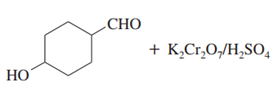
c.
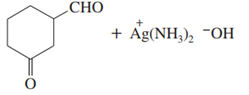
d.,

Answer
a.

b.

c,

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
Hydration of alkynes (via oxymercuration) gives good yields of single compounds only with symmetrical or asymmetrical alkynes. Show what products would be from hydration fo each compound.
(a) hex-3-yne
(b) hex-2-yne
(c) hex-1-yne
(d) cyclodecyne
(e) 3-methylcyclodecyne
Show what alcohols and carbonyl compounds give the following derivatives.
(a)
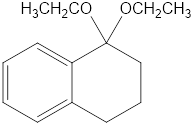
(b)
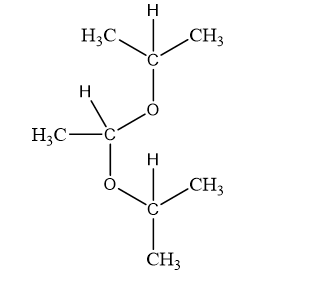
(c)
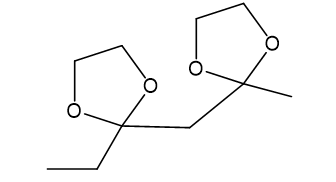
(d)
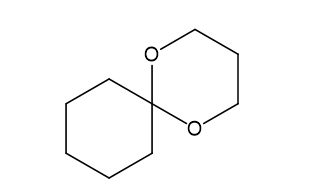
(e)
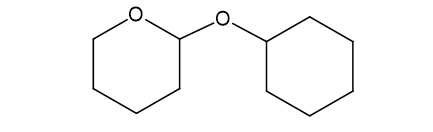
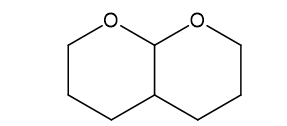
(f)
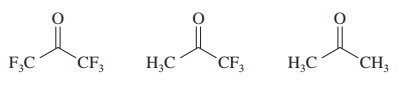
Within each set of structures, indicate which will react fastest, and which slowest, toward nucleophilic addition in basic conditions.
(a)

(b)
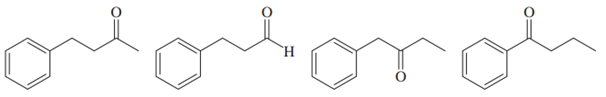
(c)
Question: Show a complete mechanism for this equilibrium established in diethyl ether with HCI gas as catalyst.
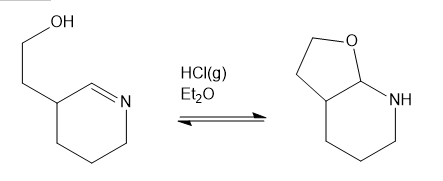
Propose a mechanism for both parts of the Wolff-Kishner reduction of cyclohexanone: the formation of the hydrazone, and then the base catalyzed reduction with evolution of nitrogen gas.
What do you think about this solution?
We value your feedback to improve our textbook solutions.