Chapter 22: Q.69-C (page 1204)
Question: Predict the products of the following reactions
c.
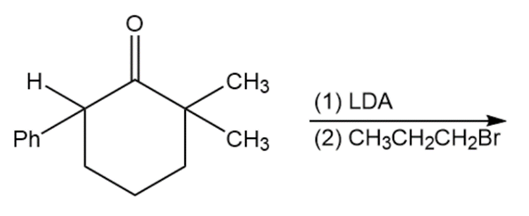
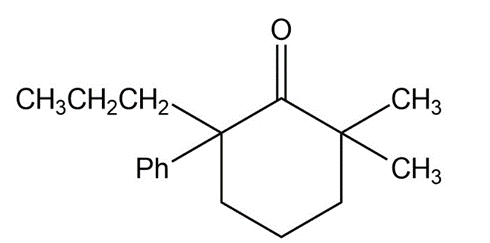
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 22: Q.69-C (page 1204)
Question: Predict the products of the following reactions
c.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Without looking back, propose a mechanism for the hydrolysis of this iminium salt to the alkylated ketone. The first step is attack by water, followed by loss of a proton to give a carbinolamine. Protonation on nitrogen allows pyrrolidine to leave, giving the protonated ketone.
Propose a mechanism for the crossed Claisen condensation between ethyl acetate and ethyl benzoate.
Phenylacetone can form two different enols.
Question:Show how you would use the Robinson annulation to synthesize the following compounds
A student wanted to dry some diacetone alcohol and allowed it to stand over anhydrous potassium carbonate for a week. At the end of the week, the sample was found to contain nearly pure acetone. Propose a mechanism for the reaction that took place.
What do you think about this solution?
We value your feedback to improve our textbook solutions.