Chapter 22: Q,67P-B (page 1204)
Question:Show how you would use the Robinson annulation to synthesize the following compounds
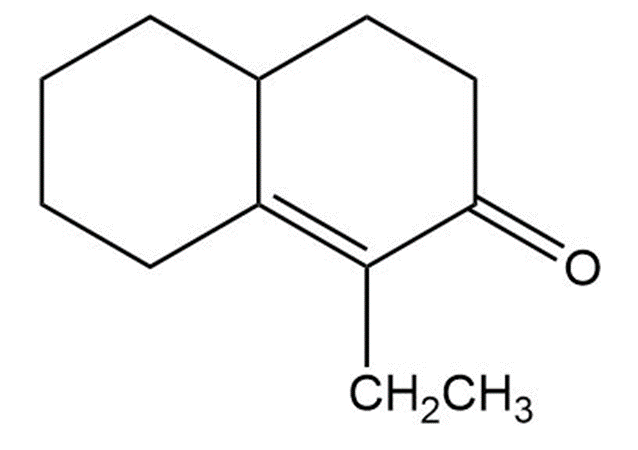
Short Answer
Answer
The starting reactants will be:
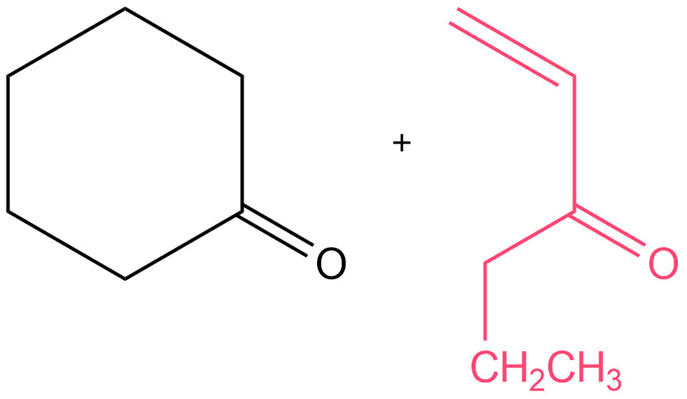
Learning Materials
Features
Discover
Chapter 22: Q,67P-B (page 1204)
Question:Show how you would use the Robinson annulation to synthesize the following compounds

Answer
The starting reactants will be:

All the tools & learning materials you need for study success - in one app.
Get started for free
When cyclodecane-1,6-dione is treated with sodium carbonate, the product gives a UV spectrum similar to that of 1-acetyl-2-methylcyclopentene. Propose a structure for the product, and give a mechanism for its formation.
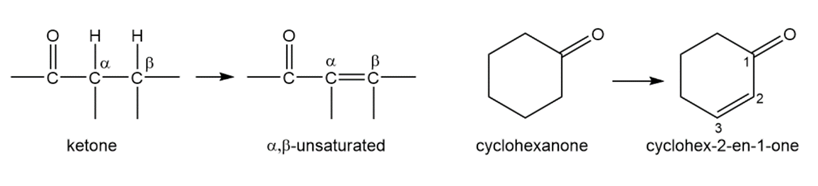
Acid-catalyzed halogenation is synthetically useful for converting ketones to α,β-unsaturated ketones, which are useful in Michael reactions (Section 22-18). Propose a method for converting cyclohexanone to cyclohex-2-en-1-one, an important synthetic starting material.
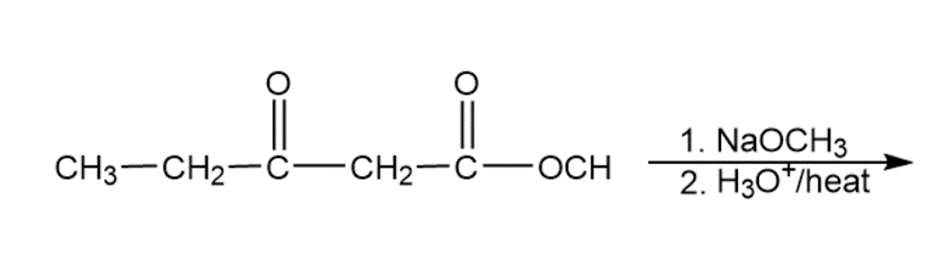
Predict the major products of the following reactions.
a)
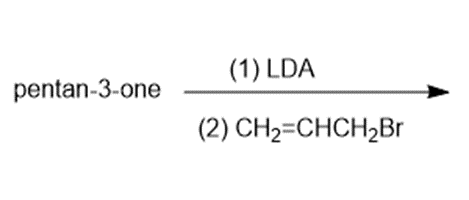
Question: Predict the products of the following reactions
h.
Phenylacetone can form two different enols.
What do you think about this solution?
We value your feedback to improve our textbook solutions.