Chapter 22: Q.67P_A (page 1204)
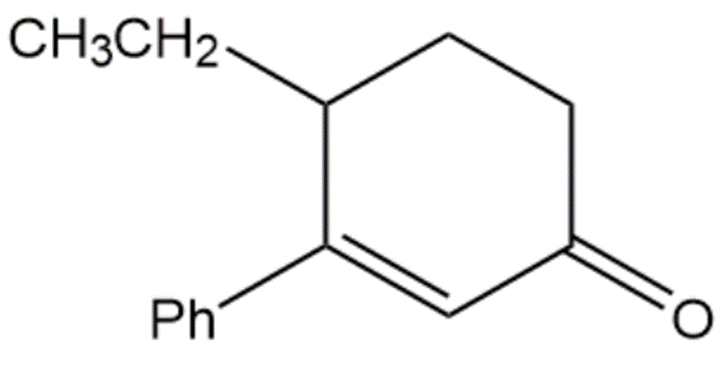
Question:Show how you would use the Robinson annulation to synthesize the following compounds.
C.
Short Answer
Answer
The starting reactants will be:

Learning Materials
Features
Discover
Chapter 22: Q.67P_A (page 1204)
Question:Show how you would use the Robinson annulation to synthesize the following compounds.
C.

Answer
The starting reactants will be:

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Predict the products of the following reactions
a. Cyclopentanone +Br2 in acetic acid
Propose mechanisms for the following reactions.
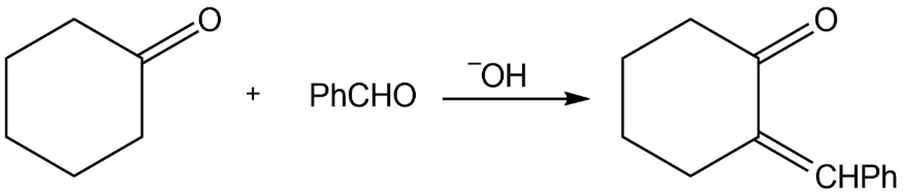
Show how you would use the acetoacetic ester synthesis to make the following compounds
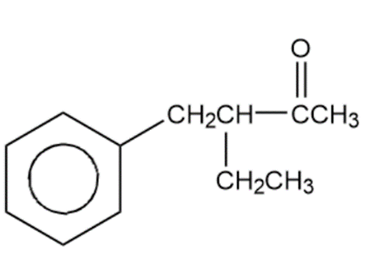
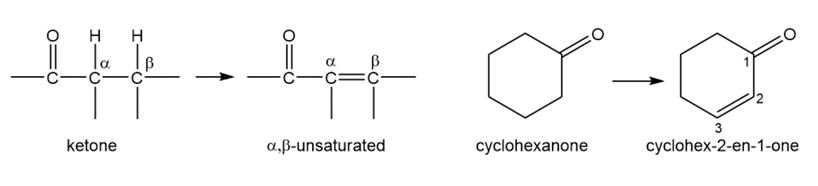
Acid-catalyzed halogenation is synthetically useful for converting ketones to α,β-unsaturated ketones, which are useful in Michael reactions (Section 22-18). Propose a method for converting cyclohexanone to cyclohex-2-en-1-one, an important synthetic starting material.
Many of the condensations we have studied are reversible. The reverse reactions are often given the prefix retro-, the Latin word meaning “backward.” Propose mechanisms to account for the following reactions.
a.
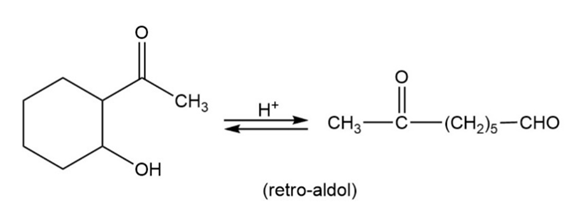
b.
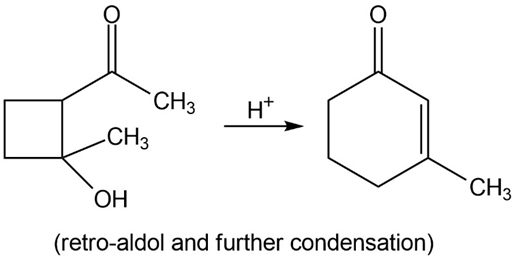
c.

d.

What do you think about this solution?
We value your feedback to improve our textbook solutions.