Chapter 22: Q68P-F (page 1204)
Question:Show how you would use an aldol, Claisen, or another type of condensation to make each compound
f.
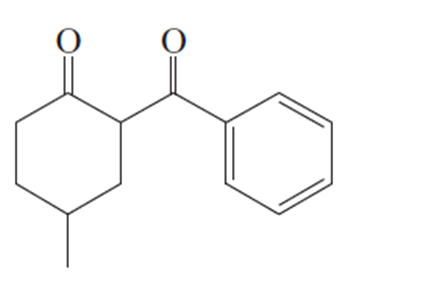
Short Answer
Answer
Two methods to obtain product:
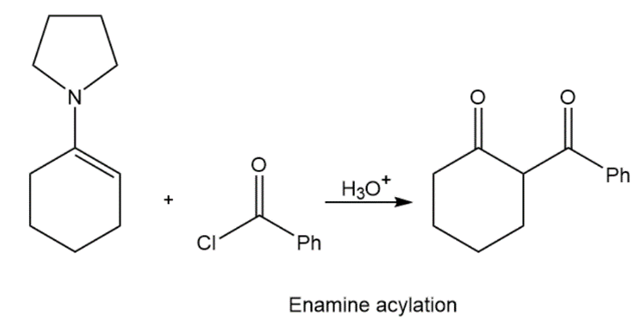
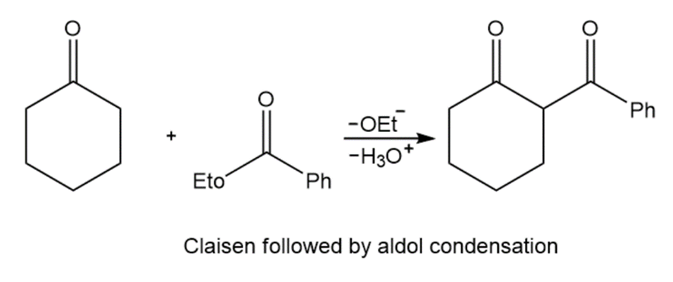
Learning Materials
Features
Discover
Chapter 22: Q68P-F (page 1204)
Question:Show how you would use an aldol, Claisen, or another type of condensation to make each compound
f.

Answer
Two methods to obtain product:


All the tools & learning materials you need for study success - in one app.
Get started for free
Predict the major products of the following reactions.
a)
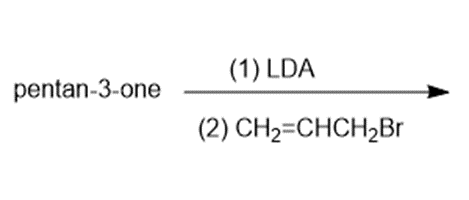
Question: Predict the products of the following reactions
e.
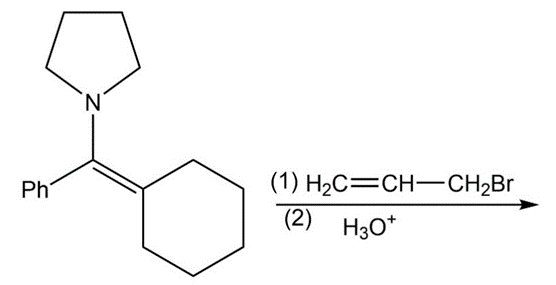
Write equations showing the expected products of the following enamine alkylation and acylation reactions. Then give the final products expected after hydrolysis of the iminium salts.
(a) pyrrolidine enamine of pentan-3-one + allyl chloride
(b) pyrrolidine enamine of acetophenone + butanoyl chloride
(c) piperidine enamine of cyclopentanone + methyl iodide
(d) piperidine enamine of cyclopentanone + methyl vinyl ketone
Question:Show how you would use an aldol, Claisen, or another type of condensation to make each compound
e.
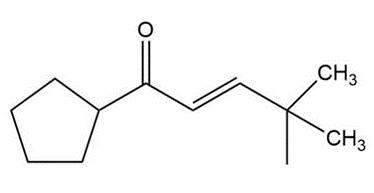
The following compounds can be synthesized by aldol condensations, followed by further reactions. (In each case, work backward from the target molecule to an aldol product, and show what compounds are needed for the condensation.)
What do you think about this solution?
We value your feedback to improve our textbook solutions.