Chapter 22: Q68P-e (page 1204)
Question:Show how you would use an aldol, Claisen, or another type of condensation to make each compound
e.
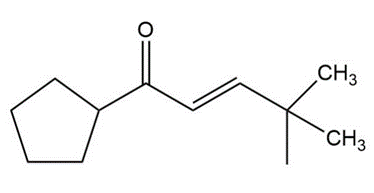
Short Answer
Answer
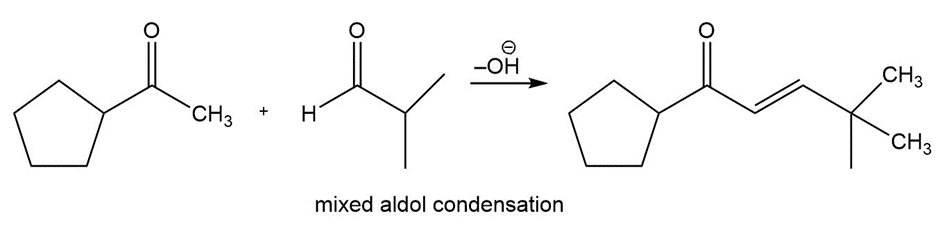
Learning Materials
Features
Discover
Chapter 22: Q68P-e (page 1204)
Question:Show how you would use an aldol, Claisen, or another type of condensation to make each compound
e.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
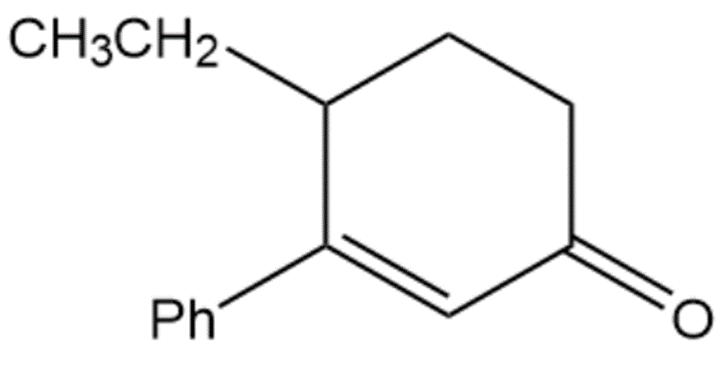
Question:Show how you would use the Robinson annulation to synthesize the following compounds.
C.
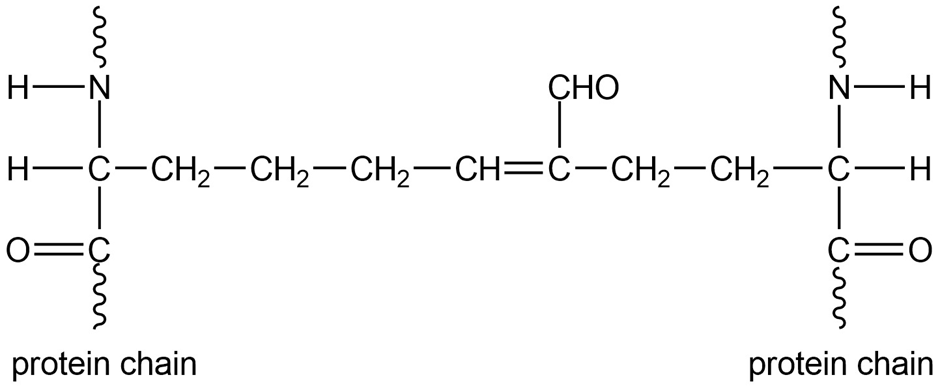
Biochemists studying the structure of collagen (a fibrous protein in connective tissue) found cross-links containing unsaturated aldehydes between protein chains. Show the structures of the side chains that react to form these crosslinks, and propose a mechanism for their formation in a weakly acidic solution.
Propose a mechanism for the acid-catalyzed reaction of cyclohexanone with pyrrolidine.
Question:Show how you would use an aldol, Claisen, or another type of condensation to make each compound
d.
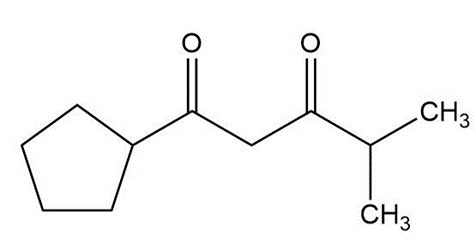
The Knoevenagel condensation is a special case of the aldol condensation in which an active methylene compound reacts with an aldehyde or ketone, in the presence of a secondary amine as a basic catalyst, to produce a new C=C. Show the starting materials that made each of these by a Knoevenagel condensation.
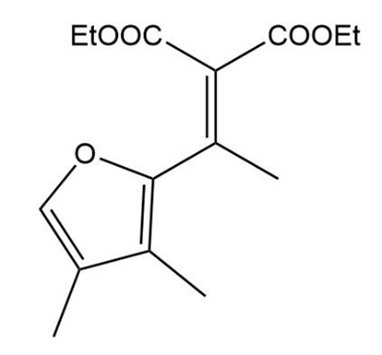
What do you think about this solution?
We value your feedback to improve our textbook solutions.