Chapter 22: Q68P-D (page 1204)
Question:Show how you would use an aldol, Claisen, or another type of condensation to make each compound
d.
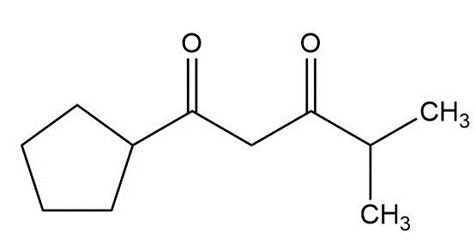
Short Answer
Answer
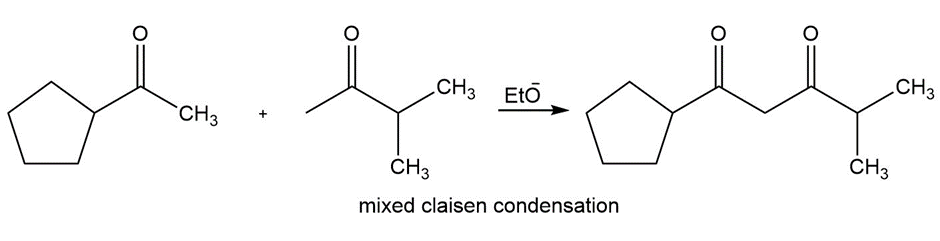
Learning Materials
Features
Discover
Chapter 22: Q68P-D (page 1204)
Question:Show how you would use an aldol, Claisen, or another type of condensation to make each compound
d.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Predict the products of these reaction sequences.
a.
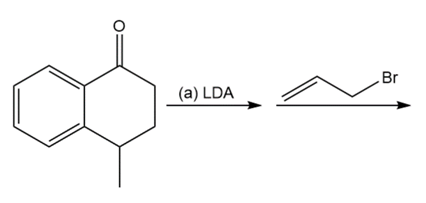
Propose a mechanism for the conjugate addition of a nucleophile (Nuc:-) to acrylonitrile (H2C=CHCN) and to nitroethylene. Use resonance forms to show how the cyano and nitro groups activate the double bond toward conjugate addition.
Question: Predict the products of the following reactions
i.
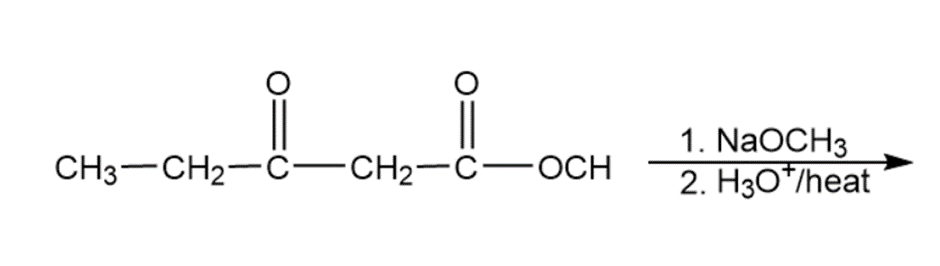
Show how you would use the malonic ester synthesis to make the following compounds
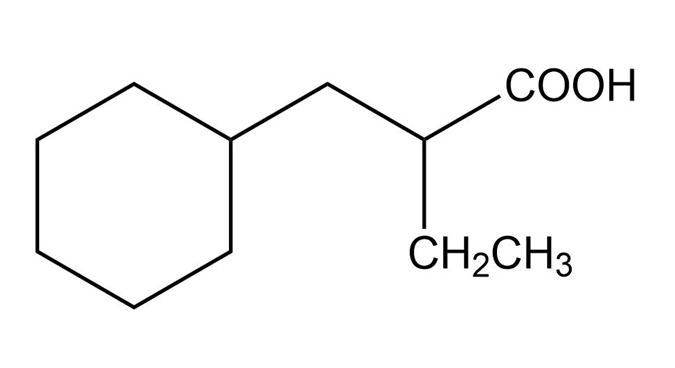
Propose a mechanism for the acid-catalyzed bromination of pentan-3-one.
What do you think about this solution?
We value your feedback to improve our textbook solutions.