Chapter 22: Q15P (page 1163)
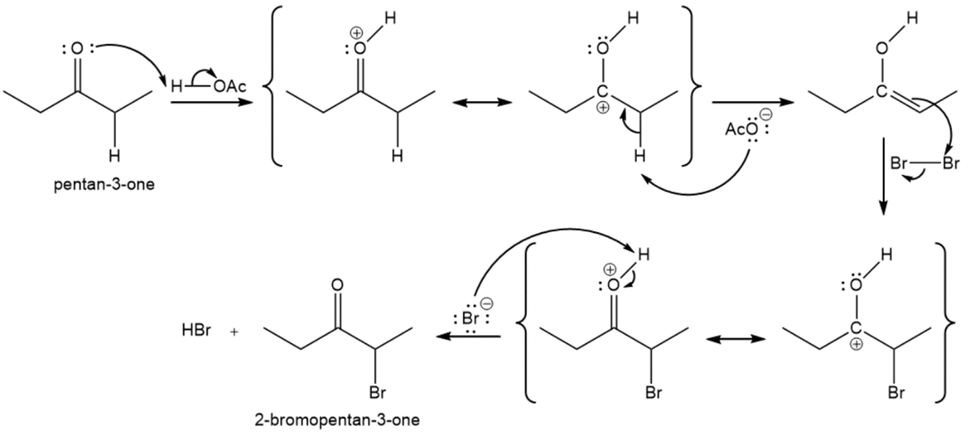
Propose a mechanism for the acid-catalyzed bromination of pentan-3-one.
Short Answer
Learning Materials
Features
Discover
Chapter 22: Q15P (page 1163)
Propose a mechanism for the acid-catalyzed bromination of pentan-3-one.

All the tools & learning materials you need for study success - in one app.
Get started for free
Show how an acetoacetic ester synthesis might be used to form a δ -diketone such as heptane-2,6-dione.
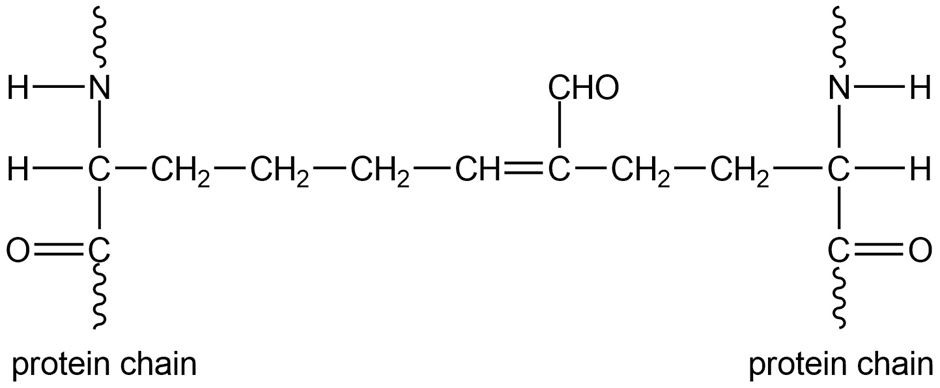
Biochemists studying the structure of collagen (a fibrous protein in connective tissue) found cross-links containing unsaturated aldehydes between protein chains. Show the structures of the side chains that react to form these crosslinks, and propose a mechanism for their formation in a weakly acidic solution.
Predict the products of the following reactions.
a. Cyclopentanone +Br2 in acetic acid
b. 1phenylethanol + I2 excess
c.
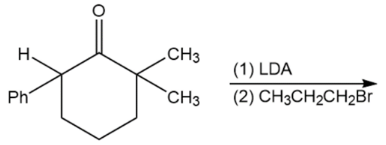
d)
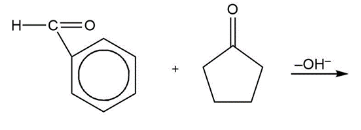
e)
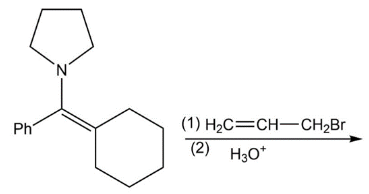
f)
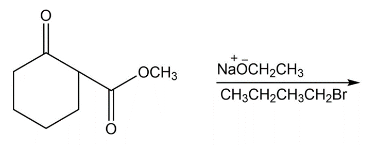
g)
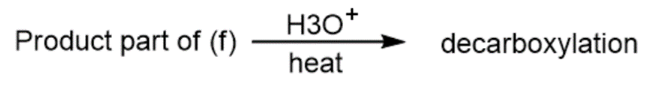
h)
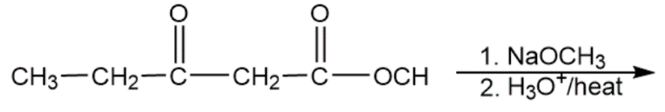
i)
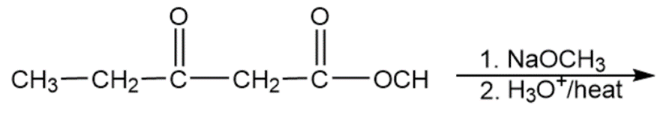
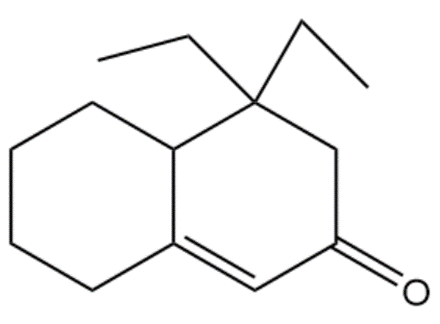
Question:Show how you would use the Robinson annulation to synthesize the following compounds.
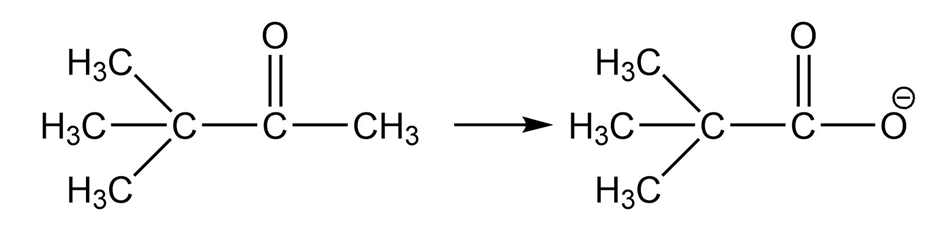
Show how you would accomplish the following conversions in good yields. You may use any necessary reagents.
What do you think about this solution?
We value your feedback to improve our textbook solutions.