Chapter 22: Q54P (page 1193)
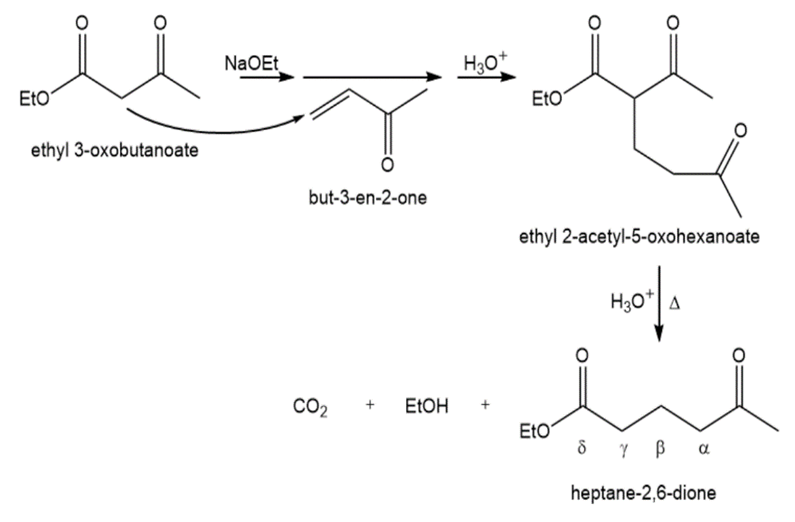
Show how an acetoacetic ester synthesis might be used to form a δ -diketone such as heptane-2,6-dione.
Short Answer
Learning Materials
Features
Discover
Chapter 22: Q54P (page 1193)
Show how an acetoacetic ester synthesis might be used to form a δ -diketone such as heptane-2,6-dione.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Predict the products of the following reactions
d.
Without looking back, propose a mechanism for the hydrolysis of this iminium salt to the alkylated ketone. The first step is attack by water, followed by loss of a proton to give a carbinolamine. Protonation on nitrogen allows pyrrolidine to leave, giving the protonated ketone.
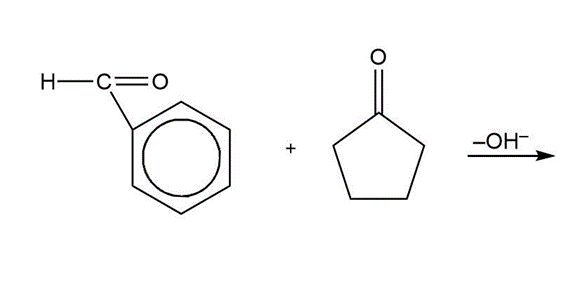
Show how cyclohexanone might be converted to the following - diketone .
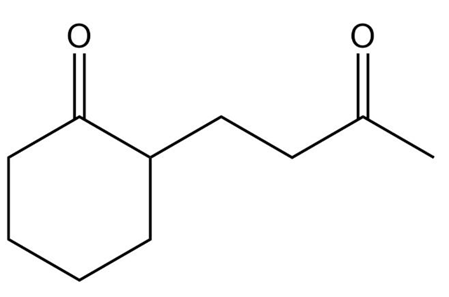
The Knoevenagel condensation is a special case of the aldol condensation in which an active methylene compound reacts with an aldehyde or ketone, in the presence of a secondary amine as a basic catalyst, to produce a new C=C. Show the starting materials that made each of these by a Knoevenagel condensation.
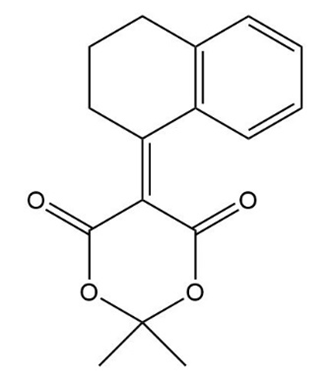
The following compound results from base-catalyzed aldol cyclization of a 2-substituted cyclohexanone.
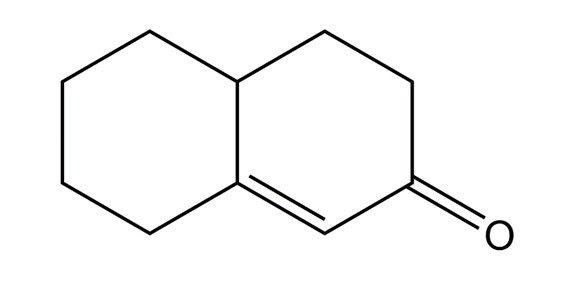
What do you think about this solution?
We value your feedback to improve our textbook solutions.