Chapter 22: Q70P-A (page 1204)
Question: Predict the products of these reaction sequences.
a.
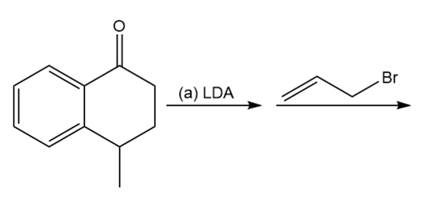
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 22: Q70P-A (page 1204)
Question: Predict the products of these reaction sequences.
a.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Predict the products of the following reactions
f.
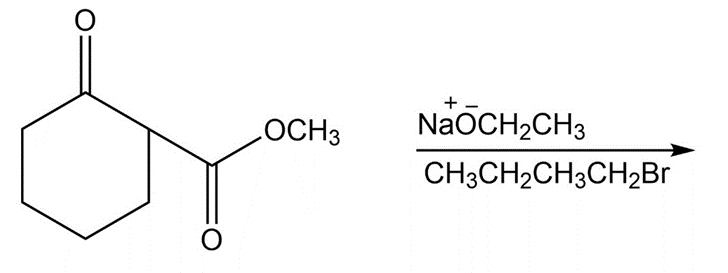
Propose mechanisms for the two Dieckmann condensations just shown.
Show the products of the reactions of these carboxylic acids with before and after hydrolysis.
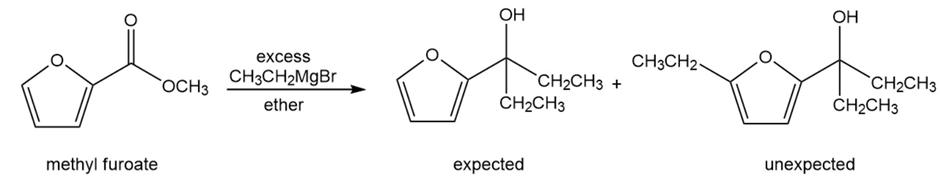
(A true story.) Chemistry lab students added an excess of ethyl magnesiumbromide to methyl furoate, expecting the Grignard reagent to add twice and form the tertiary alcohol. After water workup, they found that the product was a mixture of two compounds. One was the expected product having two ethyl groups, but the unexpected product had added three ethyl groups. Propose a mechanism to explain the formation of the unexpected product.
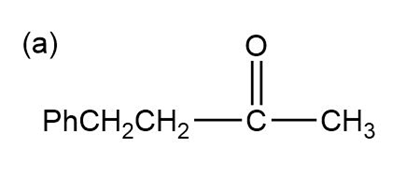
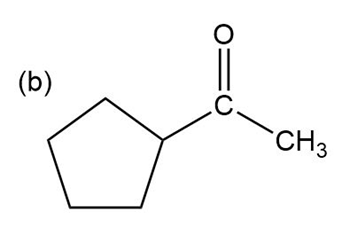
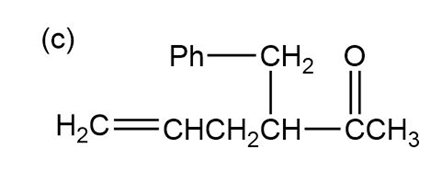
Show how the following ketones might be synthesized by using the acetoacetic ester synthesis.
What do you think about this solution?
We value your feedback to improve our textbook solutions.