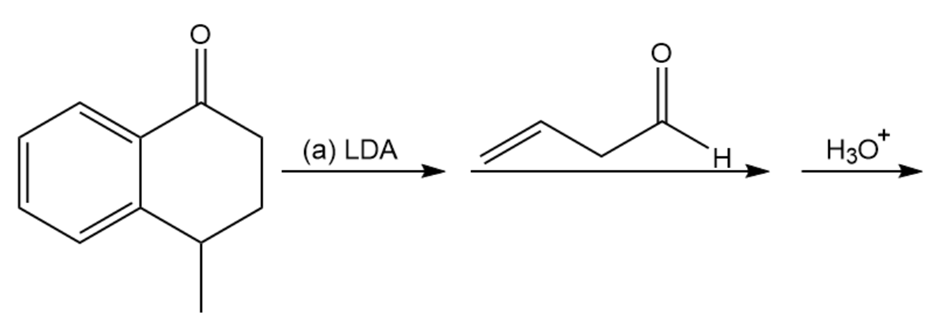
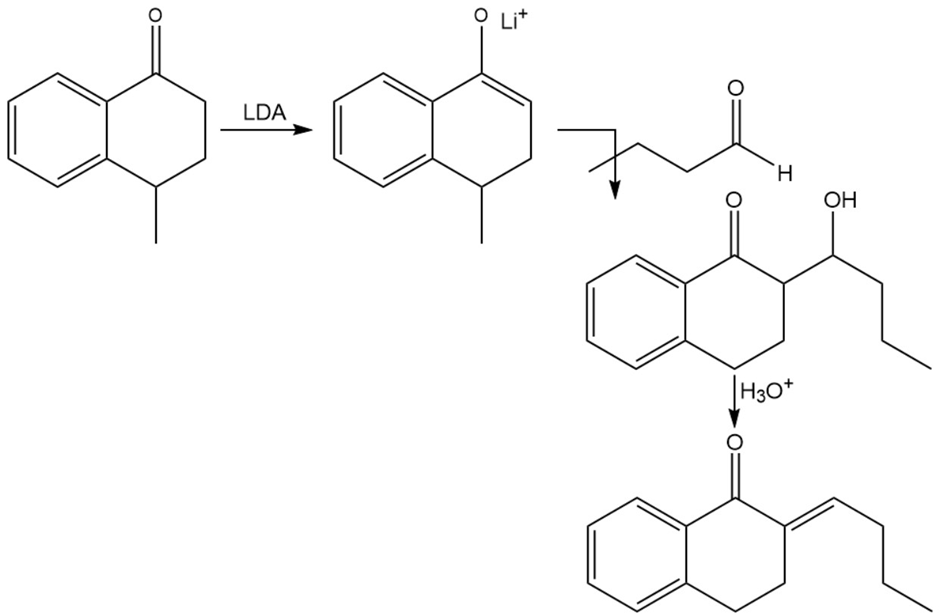
Chapter 22: Q70P-b (page 1205)
Predict the products of these reaction sequences.
Short Answer
Learning Materials
Features
Discover
Chapter 22: Q70P-b (page 1205)
Predict the products of these reaction sequences.


All the tools & learning materials you need for study success - in one app.
Get started for free
Without looking back, propose a mechanism for the hydrolysis of this iminium salt to the alkylated ketone. The first step is attack by water, followed by loss of a proton to give a carbinolamine. Protonation on nitrogen allows pyrrolidine to leave, giving the protonated ketone.
Propose a mechanism for the crossed Claisen condensation between ethyl acetate and ethyl benzoate.
Show how octane-2,7-dione might cyclize to a cycloheptenone. Explain why ring closure to the cycloheptenone is not favored.
Predict the products from this sequence of reactions
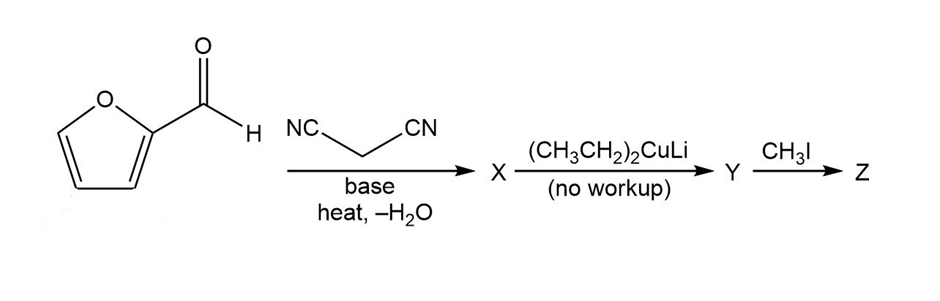
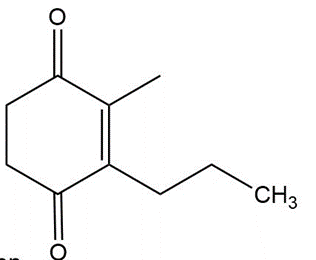
Question:Show how you would use an aldol, Claisen, or another type of condensation to make each compound
c.
What do you think about this solution?
We value your feedback to improve our textbook solutions.