Chapter 22: Q30P (page 1173)
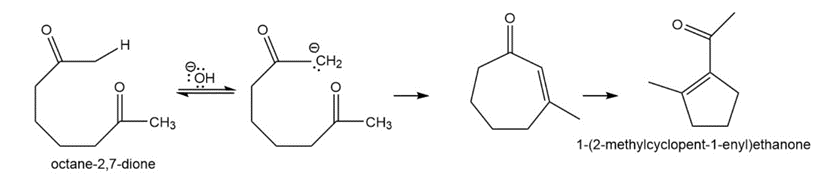
Show how octane-2,7-dione might cyclize to a cycloheptenone. Explain why ring closure to the cycloheptenone is not favored.
Short Answer
Learning Materials
Features
Discover
Chapter 22: Q30P (page 1173)
Show how octane-2,7-dione might cyclize to a cycloheptenone. Explain why ring closure to the cycloheptenone is not favored.

All the tools & learning materials you need for study success - in one app.
Get started for free
Propose mechanisms for the following reactions.
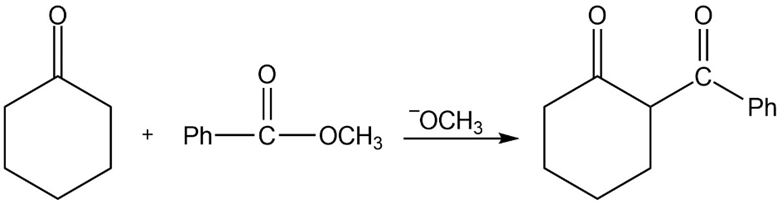
Question: Predict the products of the following reactions
i.
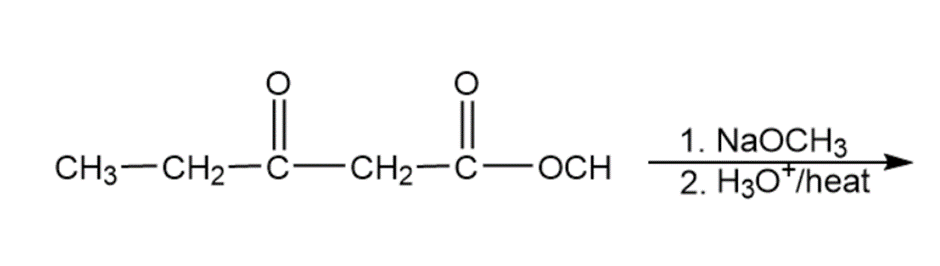
When propionaldehyde is warmed with sodium hydroxide, one of the products is 2-methylpent-2-enal. Propose a mechanism for this reaction.
Show how you would use the acetoacetic ester synthesis to make the following compounds
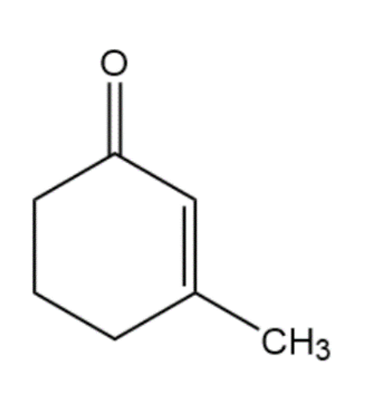
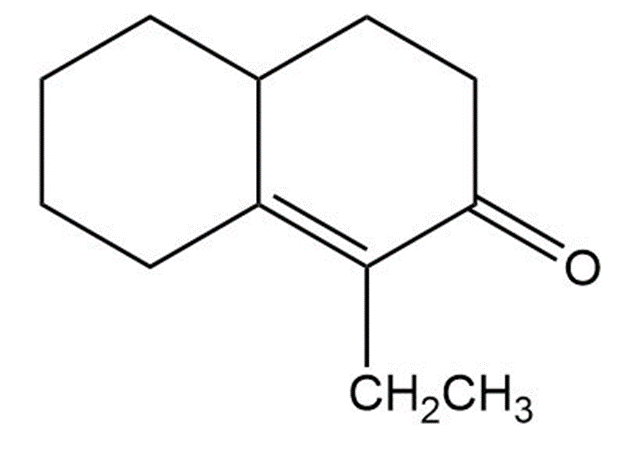
Question:Show how you would use the Robinson annulation to synthesize the following compounds
What do you think about this solution?
We value your feedback to improve our textbook solutions.