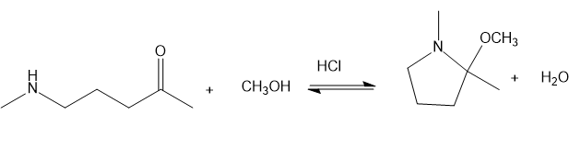
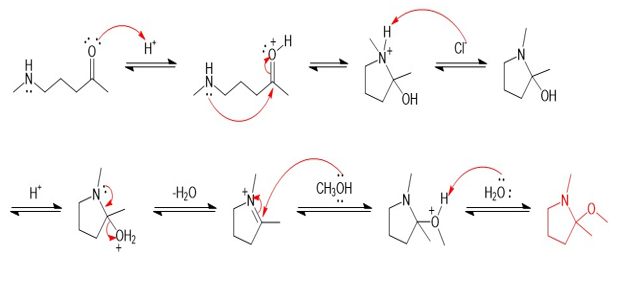
Chapter 20: Q70P (page 1038)
Show a complete mechanism for this reaction.
Short Answer
Learning Materials
Features
Discover
Chapter 20: Q70P (page 1038)
Show a complete mechanism for this reaction.


All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Show how the following ketones might be synthesized from the indicated acids, using any necessary reagents.
(a)propiophenone from propionic acid (two ways, using alkylation of the acid and using Friedel-Crafts acylation)
(b)methyl cyclohexyl ketone from cyclohexanecarboxylic acid
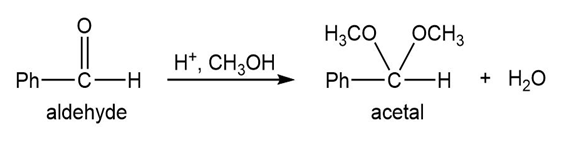
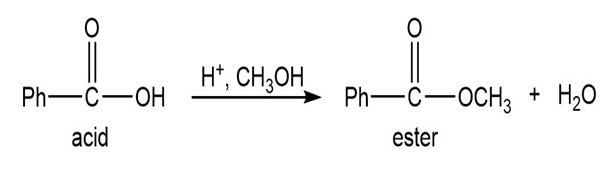
Q.13Most of the Fischer esterification mechanism is identical with the mechanism of acetal formation. The difference is in the final step, where a resonance-stabilized carbocation loses a proton to give the ester. Write mechanisms for the following reactions, with the comparable steps directly above and below each other. Explain why the final step of the esterification (proton loss) cannot occur in acetal formation, and show what happens instead.
Show how to synthesize the following compounds, using appropriate carboxylic acids and amines.
(a)
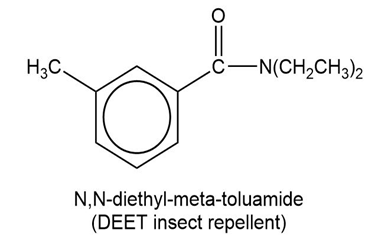
(b)
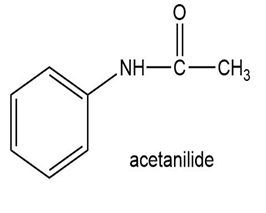
(c)
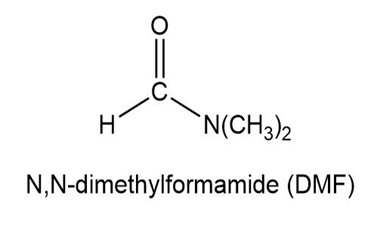
Explain why the acid-catalyzed condensation is a poor method for the synthesis of an unsymmetrical ether such as ethyl methyl ether, CH3CH2 - O - CH3.
For each compound,
(a)
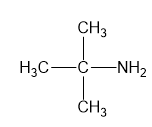
(b)
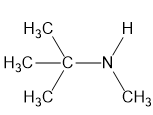
(c)
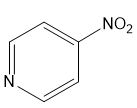
(d)
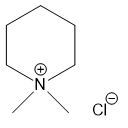
(e)
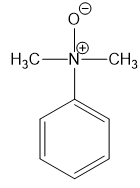
(f)
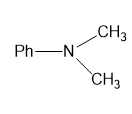
(g)
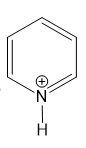
(h)
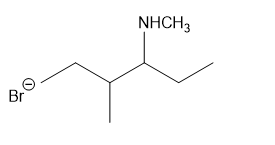
What do you think about this solution?
We value your feedback to improve our textbook solutions.